Lactitol
 | |
| Names | |
|---|---|
| Trade names | Importal, Pizensy, Lacty, others |
| Other names | Lactit, lactositol, lactobiosit[1], Lactitol hydrate (JAN JP) |
| |
| Clinical data | |
| Drug class | Osmotic laxative[2] |
| Main uses | Chronic idiopathic constipation (CIC)[2] |
| Side effects | Flatulence, diarrhea, abdominal distension, increased blood pressure[2] |
| Pregnancy category |
|
| Routes of use | By mouth |
| Legal | |
| License data | |
| Legal status |
|
| Chemical and physical data | |
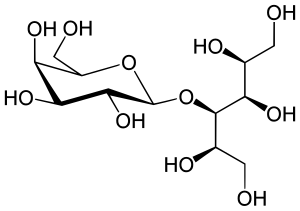
| Formula | C12H24O11 |
| Molar mass | 344.313 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 146 °C (295 °F) |
| |
| |
Lactitol is a sugar alcohol and laxative.[1] As a laxative it is used for chronic constipation of unknown cause.[2] It is taken by mouth.[2] It is also used as a replacement sweetener in low calorie foods.[1]
Common side effects include flatulence, diarrhea, abdominal distension, and increased blood pressure.[2] It is an osmotic laxative and works by pulling water into the small intestine.[2] It has about 30–40% of the sweetness of sucrose.[1]
Lactitol was first described in 1920 by Senderens.[1] It was approved for medical use in the United States in 2020.[2] It is generally recognized as safe in the United States and allowed as a sweetener in Europe.[1] Commercially it was sold for about 2.5 USD per kilogram in 2009.[3] It is made from lactose.[1]
Uses
Lactitol is used in a variety of low food energy or low fat foods. High stability makes it popular for baking. It is used in sugar-free candies, cookies (biscuits), chocolate, and ice cream, with a sweetness of 30–40% that of sucrose.[4] Lactitol also promotes colon health as a prebiotic. Because of poor absorption, lactitol only has 2–2.5 kilocalories (8.4–10.5 kilojoules) per gram,[4] compared to 4 kilocalories (17 kJ) per gram for typical saccharides. Hence, lactitol is about 60% as caloric as typical saccharides.
Medical
Lactitol is a laxative and is used to prevent or treat constipation.[5] e.g., under the trade name Importal.[6][7]
Lactitol is also an excipient in some prescription drugs.[8][9]
Side effects
Lactitol, erythritol, sorbitol, xylitol, mannitol, and maltitol are all sugar alcohols. The U.S. Food and Drug Administration (FDA) classifies sugar alcohols as "generally recognized as safe" (GRAS). They are approved as food additives, and are recognized as not contributing to tooth decay or causing increases in blood glucose. Lactitol is also approved for use in foods in most countries around the world.
Like other sugar alcohols, lactitol causes cramping, flatulence, and diarrhea in some individuals who consume it. This is because humans lack a suitable beta-galactosidase in the upper gastrointestinal (GI) tract, and a majority of ingested lactitol reaches the large intestine,[10] where it then becomes fermentable to gut microbes (prebiotic) and can pull water into the gut by osmosis. Those with health conditions should consult their GP or dietician prior to consumption.
History
In 2020, lactitol was approved for use in the United States as an osmotic laxative for the treatment of chronic idiopathic constipation (CIC) in adults.[11][12][13]
The U.S. Food and Drug Administration (FDA) approved lactitol based on evidence from a clinical trial (Trial 1/ NCT02819297) of 594 subjects with CIC conducted in the United States.[13] The FDA also considered other supportive evidence including data from Trial 2 (NCT02481947) which compared lactitol to previously approved drug (lubiprostone) for CIC, and Trial 3 (NCT02819310) in which subjects used lactitol for one year as well as data from published literature.[13]
The benefit and side effects of lactitol were evaluated in a clinical trial (Trial 1) of 594 subjects with CIC.[13] In this trial, subjects received treatment with either lactitol or placebo once daily for 6 months.[13] Neither the subjects nor the health care providers knew which treatment was being given until after the trials were completed.[13]
In the second trial (Trial 2) of three months duration, improvement in CSBMs was used to compare lactitol to the drug lubiprostone which was previously approved for CIC.[13] The third trial (Trial 3) was used to collect the side effects in subjects treated with lactitol for one year.[13]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 O'Brien-Nabors, Lyn (8 June 2001). Alternative Sweeteners, Third Edition, Revised and Expanded. CRC Press. p. 297. ISBN 978-0-8247-0437-7. Archived from the original on 12 November 2022. Retrieved 4 November 2022.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 "DailyMed - PIZENSY- lactitol powder, for solution". dailymed.nlm.nih.gov. Archived from the original on 25 October 2020. Retrieved 4 November 2022.
- ↑ Illanes, Andrés; Guerrero, Cecilia; Vera, Carlos; Wilson, Lorena; Conejeros, Raúl; Scott, Felipe (6 July 2016). Lactose-Derived Prebiotics: A Process Perspective. Academic Press. p. 100. ISBN 978-0-12-802745-5. Archived from the original on 12 November 2022. Retrieved 4 November 2022.
- ↑ 4.0 4.1 Gränzle, M.G. (2011). "Lactose and Oligosaccharides | Lactose: Derivatives". Encyclopedia of Dairy Sciences (2nd ed.). Elsevier Ltd. pp. 202–208. doi:10.1016/B978-0-12-374407-4.00275-2. ISBN 9780123744074. Archived from the original on 2022-06-15. Retrieved 2022-04-04.
- ↑ Miller LE, Tennilä J, Ouwehand AC (2014). "Efficacy and tolerance of lactitol supplementation for adult constipation: a systematic review and meta-analysis". Clin Exp Gastroenterol. 7: 241–8. doi:10.2147/CEG.S58952. PMC 4103919. PMID 25050074.
- ↑ "Importal". Drugs.com. 3 February 2020. Archived from the original on 25 February 2020. Retrieved 24 February 2020.
- ↑ FASS.se Archived 2012-02-19 at the Wayback Machine (the Swedish Medicines Information Engine). Revised 2003-02-12.
- ↑ "Lactitol (Inactive Ingredient)". Drugs.com. 23 September 2018. Archived from the original on 25 February 2020. Retrieved 24 February 2020.
- ↑ "Lactitol Monohydrate (Inactive Ingredient)". Drugs.com. 3 October 2018. Archived from the original on 25 February 2020. Retrieved 24 February 2020.
- ↑ Grimble GK, Patil DH, Silk DB (1988). "Assimilation of lactitol, an unabsorbed disaccharide in the normal human colon". Gut. 29 (12): 1666–1671. doi:10.1136/gut.29.12.1666. PMC 1434111. PMID 3220306.
- ↑ "Pizensy: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Archived from the original on 18 January 2021. Retrieved 24 February 2020.
- ↑ "Pizensy- lactitol powder, for solution". DailyMed. 21 February 2020. Archived from the original on 25 October 2020. Retrieved 24 February 2020.
- ↑ 13.0 13.1 13.2 13.3 13.4 13.5 13.6 13.7 "Drug Trial Snapshot: Pizensy". U.S. Food and Drug Administration (FDA). 12 February 2020. Archived from the original on 28 September 2020. Retrieved 4 March 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- Pages using duplicate arguments in template calls
- Webarchive template wayback links
- Wikipedia articles incorporating the PD-notice template
- Infobox drug with local INN variant
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Articles containing unverified chemical infoboxes
- Disaccharides
- E-number additives
- Laxatives
- Sugar alcohols
- Sugar substitutes
- RTT