Klebsiella pneumoniae
| Klebsiella infection | |
|---|---|
| Other names: Klebsiella[1] | |
 | |
| K. pneumoniae on a agar plate | |
| Specialty | Infectious diseases |
| Symptoms | Characteristic sputum,fever, nausea, tachycardia, and vomiting |
| Types | Hypervirulent Klebsiella pneumonia, Klebsiella pneumonia |
| Causes | K. pneumoniae must enter the respiratory tract to cause pneumoniae |
| Diagnostic method | CBC,Sputum(culture), Radiography(chest),CT scan |
| Treatment | pneumonia is by antibiotics like Aminoglycosides and Cephalosporins |
| Klebsiella pneumoniae | |
|---|---|
| Scientific classification | |
| Domain: | Bacteria |
| Phylum: | Pseudomonadota |
| Class: | Gammaproteobacteria |
| Order: | Enterobacterales |
| Family: | Enterobacteriaceae |
| Genus: | Klebsiella |
| Species: | K. pneumoniae
|
| Binomial name | |
| Klebsiella pneumoniae (Schroeter 1886) Trevisan 1887
| |
| Subspecies | |
Klebsiella pneumoniae is a Gram-negative, non-motile, encapsulated, lactose-fermenting, facultative anaerobic, rod-shaped bacterium. It appears as a mucoid lactose fermenter on MacConkey agar. Although found in the normal flora of the mouth, skin, and intestines,[2] it can cause destructive changes to human and animal lungs if aspirated, specifically to the alveoli resulting in bloody, brownish or yellow colored jelly like sputum. In the clinical setting, it is the most significant member of the genus Klebsiella of the Enterobacteriaceae. K. oxytoca and K. rhinoscleromatis have also been demonstrated in human clinical specimens. In recent years, Klebsiella species have become important pathogens in nosocomial infections.[3][4][5]
It naturally occurs in the soil, and about 30% of strains can fix nitrogen in anaerobic conditions.[6] As a free-living diazotroph, its nitrogen-fixation system has been much-studied, and is of agricultural interest, as K. pneumoniae has been demonstrated to increase crop yields in agricultural conditions.[7]
It is closely related to K. oxytoca from which it is distinguished by being indole-negative and by its ability to grow on melezitose.[8]
Forms (types)
- Hypervirulent Klebsiella pneumonia
It is a very virulent strain coming from Asia, with a high mortality rate. It often spreads to central nervous system and eye (causing endophthalmitis). A string test is used to help the diagnosis.[9] Further examinations and treatments are made on a case-by-case basis, as there are currently no international guidelines.[10]
- Klebsiella pneumonia
The most common condition caused by Klebsiella bacteria outside the hospital is pneumonia, typically in the form of bronchopneumonia and also bronchitis. These patients have an increased tendency to develop lung abscess, cavitation, empyema, and bronchiectasis. It has a death rate around 11-28%, even with surgical treatment.[3][11]
Signs and symptoms
Individuals with Klebsiella pneumonia tend to cough up a characteristic sputum, as well as having fever, nausea, tachycardia, and vomiting. Klebsiella pneumonia tends to affect people with underlying conditions, such as alcoholism.[12]
Pathophysiology
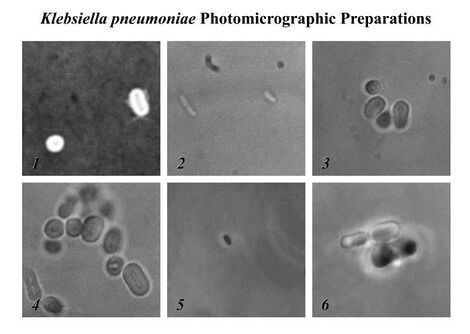
It is typically due to aspiration and alcoholism may be a risk factor, though it is also commonly implicated in hospital-acquired urinary tract infections, and COPD (chronic obstructive pulmonary disease) individuals[12][13]
In terms of the pathophysiology of Klebsiella pneumonia we see neutrophil myeloperoxidase defense against K. pneumoniae. Oxidative inactivation of elastase is involved, while LBP helps transfer bacteria cell wall elements to the cells.[14][15]
Transmission
To get a K. pneumoniae infection, a person must be exposed to the bacteria. In other words, K. pneumoniae must enter the respiratory tract to cause pneumoniae, or the blood to cause a bloodstream infection. In healthcare settings, K. pneumoniae bacteria can be spread through person-to-person contact (for example, contaminated hands of healthcare personnel, or other people via patient to patient) or, less commonly, by contamination of the environment; the role of transmission directly from the environment to patients is controversial and requires further investigation.[16] However, the bacteria are not spread through the air. Patients in healthcare settings also may be exposed to K. pneumoniae when they are on ventilators, or have intravenous catheters or wounds. These medical tools and conditions may allow K. pneumoniae to enter the body and cause infection.[17]
Resistant strains
Klebsiella organisms are often resistant to multiple antibiotics. Current evidence implicates plasmids as the primary source of the resistance genes.[18] Klebsiella species with the ability to produce extended-spectrum beta-lactamases (ESBL) are resistant to virtually all beta-lactam antibiotics, except carbapenems. Other frequent resistance targets include aminoglycosides, fluoroquinolones, tetracyclines, chloramphenicol, and trimethoprim/sulfamethoxazole.[19]

Infection with carbapenem-resistant Enterobacteriaceae (CRE) or carbapenemase-producing Enterobacteriaceae is emerging as an important challenge in health-care settings.[21][22] One of many CREs is carbapenem-resistant Klebsiella pneumoniae (CRKP). Over the past 10 years, a progressive increase in CRKP has been seen worldwide; however, this new emerging nosocomial pathogen is probably best known for an outbreak in Israel that began around 2006 within the healthcare system there.[23] In the US, it was first described in North Carolina in 1996;[24] since then CRKP has been identified in 41 states;[25] and is routinely detected in certain hospitals in New York and New Jersey.
CRKP is resistant to almost all available antimicrobial agents, and infections with CRKP have caused high rates of morbidity and mortality, in particular among persons with prolonged hospitalization and those critically ill and exposed to invasive devices; the concern is that carbapenem is often used when battling resistant bacterial strains. [3][26]
A number of mechanisms cause carbapenem resistance in the Enterobacteriaceae. These include hyperproduction of ampC beta-lactamase with an outer membrane porin mutation, CTX-M extended-spectrum beta-lactamase with a porin mutation or drug efflux, and carbapenemase production. The most important mechanism of resistance by CRKP is the production of a carbapenemase enzyme, blakpc. The gene that encodes the blakpc enzyme is carried on a mobile piece of genetic material, which increases the risk for dissemination. CRE can be difficult to detect because some strains that harbor blakpc have minimum inhibitory concentrations that are elevated, but still within the susceptible range for carbapenems. Because these strains are susceptible to carbapenems, they are not identified as potential clinical or infection control risks using standard susceptibility testing guidelines. Patients with unrecognized CRKP colonization have been reservoirs for transmission during nosocomial outbreaks.[27]
The extent and prevalence of CRKP within the environment is currently unknown. The mortality rate is also unknown, but has been observed to be as high as 44%.[28] The Centers for Disease Control and Prevention released guidance for aggressive infection control to combat CRKP:
- Place all patients colonized or infected with carbapenemase-producing Enterobacteriaceae on contact precautions. Acute-care facilities are to establish a protocol, in conjunction with the guidelines of the Clinical and Laboratory Standards Institute, to detect nonsusceptibility and carbapenemase production in Enterobacteriaceae, in particular Klebsiella spp. and Escherichia coli, and immediately alert epidemiology and infection-control staff members if identified. All acute-care facilities are to review microbiology records for the preceding 6–12 months to ensure that there have not been previously unrecognized CRE cases. If they do identify previously unrecognized cases, a point prevalence survey (a single round of active surveillance cultures) in units with patients at high risk (e.g., intensive-care units, units where previous cases have been identified, and units where many patients are exposed to broad-spectrum antimicrobials) is needed to identify any additional patients colonized with carbapenem-resistant or carbapenemase-producing Klebsiella spp. and E. coli. When a case of hospital-associated CRE is identified, facilities should conduct a round of active surveillance testing of patients with epidemiologic links to the CRE case (e.g., those patients in the same unit or patients having been cared for by the same health-care personnel).[29]
One specific example of this containment policy could be seen in Israel in 2007.[30] This policy had an intervention period from April, 2007, to May, 2008. A nationwide outbreak of CRE (which peaked in March, 2007 at 55.5 cases per 100,000 patient days) necessitated a nationwide treatment plan. The intervention entailed physical separation of all CRE carriers and appointment of a task force to oversee efficacy of isolation by closely monitoring hospitals and intervening when necessary. After the treatment plan (measured in May, 2008), the number of cases per 100,000 patient days decreased to 11.7. The plan was effective because of strict hospital compliance, wherein each was required to keep detailed documentation of all CRE carriers. In fact, for each increase in compliance by 10%, incidence of cases per 100,000 patient days decreased by 0.6.[30]
In the United States, the reasons the CDC is recommending the detection of carbapenem resistance or carbapenemase production only for Klebsiella spp. and E. coli are: this facilitates performing the test in the microbiology laboratory without the use of molecular methods, and these organisms represent the majority of CREs encountered in the United States.[31]
In mid-August 2016, a resident of Washoe County was hospitalized in Reno due to a CRE (specifically Klebsiella pneumoniae) infection. In early September of the same year, she developed septic shock and died. On testing by CDC an isolate from the patient was found to be resistant to all 26 antibiotics available in the US, including drug of last resort colistin.[32] It is believed she may have picked up the microbe while hospitalized in India for two years due to a broken right femur and subsequent femur and hip infections.[33][34][35]
Diagnosis

In terms of the diagnosis of Klebsiella pneumonia the following can be done to determine if the individual has this infection, including susceptibility testing for (ESBL) Extended Spectrum β-Lactamase, as well as:[14][12]
- CBC
- Sputum(culture)
- Radiography(chest)
- CT scan
Prevention
To prevent spreading Klebsiella infections between patients, healthcare personnel must follow specific infection-control precautions,[17] which may include strict adherence to hand hygiene (preferably using an alcohol based hand rub (60-90%) or soap and water if hands are visibly soiled. Alcohol based hand rubs are effective against these Gram-negative bacilli)[36] and wearing gowns and gloves when they enter rooms where patients with Klebsiella–related illnesses are housed. Healthcare facilities also must follow strict cleaning procedures to prevent the spread of Klebsiella.[17]
To prevent the spread of infections, patients also should clean their hands very often, including:[17]
- Before preparing or eating food
- Before touching their eyes, nose, or mouth
- Before and after changing wound dressings or bandages
- After using the restroom
- After blowing their nose, coughing, or sneezing
- After touching hospital surfaces such as bed rails, bedside tables, doorknobs, remote controls, or the phone
Treatment
K. pneumoniae can be treated with antibiotics if the infections are not drug-resistant. Infections by K. pneumoniae can be difficult to treat because fewer antibiotics are effective against them. In such cases, a microbiology laboratory must run tests to determine which antibiotics will treat the infection.[17] More specific treatments of Klebsiella pneumonia are given in its section above. For urinary tract infections with multidrug-resistant Klebsiella species, a combination therapy with amikacin and meropenem has been suggested.[37]
Treatment for Klebsiella pneumonia is by antibiotics such as aminoglycosides and cephalosporins, the choice depending upon the person's health condition, medical history and severity of the disease.[13][38]


Klebsiella possesses beta-lactamase giving it resistance to ampicillin, many strains have acquired an extended-spectrum beta-lactamase with additional resistance to carbenicillin, amoxicillin, and ceftazidime. The bacteria remain susceptible to aminoglycosides and cephalosporins, varying degrees of inhibition of the beta-lactamase with clavulanic acid have been reported. Infections due to multidrug-resistant gram-negative pathogens in the ICU have invoked the re-emergence of colistin. However, colistin-resistant strains of K. pneumoniae have been reported in ICUs.[14][39][40][41] In 2009, strains of K. pneumoniae with gene called New Delhi metallo-beta-lactamase ( NDM-1) that even gives resistance against intravenous antibiotic carbapenem, were discovered in India and Pakistan. Klebsiella cases in Taiwan have shown abnormal toxicity, causing liver abscesses in people with diabetes mellitus (DM), treatment consists of third generation cephalosporins.[42][43][44]
Epidemiology
Illness affects men with debilitating diseases; this patient population is believed to have impaired respiratory host defenses, including persons with diabetes, alcoholism, malignancy, liver disease, chronic obstructive pulmonary diseases, glucocorticoid therapy, kidney failure, and certain occupational exposures. Many of these infections are obtained when a person is in the hospital for some other reason (a nosocomial infection).[3][45][46]
In addition to pneumonia, Klebsiella can also cause infections in the urinary tract, and surgical wound sites. For patients with an invasive device in their bodies, contamination of the device becomes a risk; neonatal ward devices, respiratory support equipment, and urinary catheters put patients at increased risk.[3][45][46]
History

The genus Klebsiella was named after the German microbiologist Edwin Klebs (1834–1913).He is mainly known for his work on infectious diseases. His works paved the way for the beginning of modern bacteriology, and inspired Louis Pasteur and Robert Koch. He was the first to identify a bacterium that causes diphtheria. Klebsiella pneumoniae is also known as Friedlander's bacillum in honor of Carl Friedländer, a German pathologist, who proposed that this bacterium was the etiological factor for the pneumonia seen especially in immunocompromised individuals such as people with chronic diseases.[47][48][49][50]
Community-acquired pneumonia caused by Klebsiella pneumoniae may be called Friedländer's bacillus.[51]
Research
Research conducted at King's College, London has implicated molecular mimicry between HLA-B27 and two Klebsiella surface molecules as the cause of ankylosing spondylitis.[52]Klebsiella ranks second to E. coli for urinary tract infections in older people.[53] It is also an opportunistic pathogen for patients with chronic pulmonary disease, enteric pathogenicity, nasal mucosa atrophy, and rhinoscleroma. [54]
Multiple drug-resistant K. pneumoniae strains have been killed in vivo by intraperitoneal, intravenous, or intranasal administration of phages in laboratory tests.[55] Resistance to phages is not likely to be as troublesome as to antibiotics as new infectious phages are likely to be available in environmental reservoirs. Phage therapy can be used in conjunction with antibiotics, to supplement their activity instead of replacing it altogether.[56]
Use in research
In terms of research, the genus Klebsiella has other important species including one called Klebsiella aerogenes[57]which is often used (after being neutralized) to help Dictyostelium discoideum (which is an NIH model organism) cells grow in culture[58]
References
- ↑ "Klebsiella infection | Genetic and Rare Diseases Information Center (GARD) – an NCATS Program". rarediseases.info.nih.gov. Archived from the original on 14 September 2021. Retrieved 27 November 2021.
- ↑ Ryan, KJ; Ray, CG, eds. (2004). Sherris Medical Microbiology (4th ed.). McGraw Hill. ISBN 978-0-8385-8529-0.
- ↑ 3.0 3.1 3.2 3.3 3.4 Ashurst, John V.; Dawson, Adam (2022). "Klebsiella Pneumonia". StatPearls. StatPearls Publishing. Archived from the original on 15 May 2020. Retrieved 17 October 2022.
- ↑ Singh, Lavan; Cariappa, M.P.; Kaur, Mandeep (December 2016). "Klebsiella oxytoca: An emerging pathogen?". Medical Journal, Armed Forces India. 72 (Suppl 1): S59–S61. doi:10.1016/j.mjafi.2016.05.002. ISSN 0377-1237. Archived from the original on 3 October 2022. Retrieved 15 October 2022.
- ↑ Gundogan, N. (1 January 2014). "Klebsiella". Encyclopedia of Food Microbiology (Second Edition). Academic Press. pp. 383–388. ISBN 978-0-12-384733-1. Retrieved 15 October 2022.
- ↑ Postgate, J (1998). Nitrogen Fixation (3rd ed.). Cambridge University Press. ISBN 978-0-521-64047-3.
- ↑ Riggs, PJ; Chelius MK; Iniguez AL; Kaeppler SM; Triplett EW (2001). "Enhanced maize productivity by inoculation with diazotrophic bacteria". Australian Journal of Plant Physiology. 29 (8): 829–836. doi:10.1071/PP01045.
- ↑ Falkow, Stanley; Rosenberg, Eugene; Schleifer, Karl-Heinz; Stackebrandt, Erko (12 October 2006). The Prokaryotes: Vol. 6: Proteobacteria: Gamma Subclass. Springer Science & Business Media. p. 165. ISBN 978-0-387-25496-8. Archived from the original on 8 October 2022. Retrieved 8 October 2022.
- ↑ Hagiya, Hideharu; Watanabe, Naoto; Maki, Miyako; Murase, Tomoko; Otsuka, Fumio (2014-05-19). "Clinical utility of string test as a screening method for hypermucoviscosity‐phenotype K lebsiella pneumoniae". Acute Medicine & Surgery. 1 (4): 245–246. doi:10.1002/ams2.40. ISSN 2052-8817. PMC 5997228. PMID 29930857.
- ↑ Russo, Thomas A.; Marr, Candace M. (2019-06-19). "Hypervirulent Klebsiella pneumoniae". Clinical Microbiology Reviews. 32 (3). doi:10.1128/CMR.00001-19. ISSN 0893-8512. PMC 6589860. PMID 31092506.
- ↑ Kuhajda, Ivan; Zarogoulidis, Konstantinos; Tsirgogianni, Katerina; Tsavlis, Drosos; Kioumis, Ioannis; Kosmidis, Christoforos; Tsakiridis, Kosmas; Mpakas, Andrew; Zarogoulidis, Paul; Zissimopoulos, Athanasios; Baloukas, Dimitris; Kuhajda, Danijela (2015). "Lung abscess-etiology, diagnostic and treatment options". Annals of Translational Medicine. 3 (13): 183. doi:10.3978/j.issn.2305-5839.2015.07.08. ISSN 2305-5839. Archived from the original on 5 July 2022. Retrieved 17 October 2022.
- ↑ 12.0 12.1 12.2 "Aspiration Pneumonia Symptoms. Treatment and Information | Patient". Patient. Archived from the original on 16 January 2017. Retrieved 13 January 2017.
- ↑ 13.0 13.1 "Klebsiella species – GOV.UK". www.gov.uk. Archived from the original on 16 January 2017. Retrieved 13 January 2017.
- ↑ 14.0 14.1 14.2 Klebsiella Infections at eMedicine
- ↑ Li B, Zhao Y, Liu C, Chen Z, Zhou D (2014). "Molecular pathogenesis of Klebsiella pneumoniae". Future Microbiology. 9 (9): 1071–81. doi:10.2217/fmb.14.48. PMID 25340836.
- ↑ "Carbapenem-resistant Enterobacteriaceae (CRE) Infection: Clinician FAQs". Cdc.gov. Archived from the original on 25 October 2017. Retrieved 25 October 2017.
- ↑ 17.0 17.1 17.2 17.3 17.4 "Guideline for Isolation Precautions: Preventing Transmission of Infectious Agents in Healthcare Settings 2007". Centers for Disease Control and Prevention. 19 February 2021. Archived from the original on 19 October 2021. Retrieved 9 October 2021.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ Hudson, Corey; Bent, Zachary; Meagher, Robert; Williams, Kelly (June 6, 2014). "Resistance Determinants and Mobile Genetic Elements of an NDM-1-Encoding Klebsiella pneumoniae Strain". PLOS ONE. 9 (6): e99209. Bibcode:2014PLoSO...999209H. doi:10.1371/journal.pone.0099209. PMC 4048246. PMID 24905728.
- ↑ Nathisuwan, S; Burgess, DS; Lewis, JS (August 2001). "Extended-Spectrum β-Lactamases: Epidemiology, Detection, and Treatment". Pharmacotherapy. 21 (8): 920–928. doi:10.1592/phco.21.11.920.34529. PMID 11718498. S2CID 73938823.
- ↑ "Superbatterio New Delhi: salgono a 147 i casi in Toscana" (in italiano). Il Tirreno. 13 December 2019. Archived from the original on 14 December 2019. Retrieved 14 December 2019.
- ↑ Limbago, BM; Rasheed, JK; Anderson, KF; Zhu, W; et al. (December 2011). "IMP-Producing Carbapenem-Resistant Klebsiella pneumoniae in the United States". Journal of Clinical Microbiology. 49 (12): 4239–4245. doi:10.1128/JCM.05297-11. PMC 3233008. PMID 21998425.
- ↑ Ghaith, Doaa M.; Mohamed, Zeinat K.; Farahat, Mohamed G.; Aboulkasem Shahin, Walaa; Mohamed, Hadeel O. (March 2019). "Colonization of intestinal microbiota with carbapenemase-producing Enterobacteriaceae in paediatric intensive care units in Cairo, Egypt". Arab Journal of Gastroenterology. 20 (1): 19–22. doi:10.1016/j.ajg.2019.01.002. PMID 30733176. S2CID 73444389. Archived from the original on 2021-10-12. Retrieved 2021-10-09.
- ↑ Berrie, C (2007-04-04). "Carbapenem-resistant Klebsiella pneumoniae outbreak in an Israeli hospital". Medscape. Medical News. WebMD. Archived from the original on 2012-12-02. Retrieved 2013-07-07.
- ↑ Yigit, H; Queenan, AM; Anderson, GJ; Domenech-Sanchez, A; et al. (April 2001). "Novel carbapenem-hydrolyzing beta-lactamase, KPC-1, from a carbapenem-resistant strain of Klebsiella pneumoniae". Antimicrobial Agents and Chemotherapy. 45 (4): 1151–1161. doi:10.1128/AAC.45.4.1151-1161.2001. PMC 90438.
- ↑ Vastag, Brian (2012-08-22). "'Superbug' stalked NIH hospital last year, killing six". The Washington Post. Archived from the original on 2012-12-23. Retrieved 2013-07-07.
- ↑ Reyes, Jorge; Aguilar, Ana Cristina; Caicedo, Andrés (28 November 2019). "Carbapenem-Resistant Klebsiella pneumoniae: Microbiology Key Points for Clinical Practice". International Journal of General Medicine. 12: 437–446. doi:10.2147/IJGM.S214305. ISSN 1178-7074. Retrieved 5 October 2022.
- ↑ "Public Health Agency of Canada (PHAC) – Agence de la sante publique du Canada (ASPC)". Phac-aspc.gc.ca. 2004-09-24. Archived from the original on 2017-10-24. Retrieved 25 October 2017.
- ↑ Schwaber, Mitchell J.; Klarfeld-Lidji, Shiri; Navon-Venezia, Shiri; Schwartz, David; Leavitt, Azita; Carmeli, Yehuda (2008-03-01). "Predictors of carbapenem-resistant Klebsiella pneumoniae acquisition among hospitalized adults and effect of acquisition on mortality". Antimicrobial Agents and Chemotherapy. 52 (3): 1028–1033. doi:10.1128/AAC.01020-07. ISSN 0066-4804. PMC 2258527. PMID 18086836.
- ↑ Lledo, W; Hernandez, M; Lopez, E; Molinari, OL; et al. (2009-03-20). "Guidance for Control of Infections with Carbapenem-Resistant or Carbapenemase-Producing Enterobacteriaceae in Acute Care Facilities". Morbidity and Mortality Weekly Report. 58 (10): 256–260. PMID 19300408. Archived from the original on 2021-09-03. Retrieved 2021-10-09.
- ↑ 30.0 30.1 Schwaber, MJ; Lev, B; Israeli, A; Solter, E; et al. (2011-04-01). "Containment of a country-wide outbreak of carbapenem-resistant Klebsiella pneumoniae in Israeli hospitals via a nationally implemented intervention". Clinical Infectious Diseases. 52 (7): 848–855. doi:10.1093/cid/cir025. PMID 21317398.
- ↑ "Guidance for Control of Infections with Carbapenem-Resistant or Carbapenemase-Producing Enterobacteriaceae in Acute Care Facilities". www.cdc.gov. Archived from the original on 3 September 2021. Retrieved 8 October 2022.
- ↑ Gallagher, James (13 January 2017). "Bug resistant to all antibiotics kills woman". BBC News. Archived from the original on 16 January 2017. Retrieved 16 January 2017.
- ↑ "Nevada woman dies of superbug resistant to all available US antibiotics". STAT. 12 January 2017. Archived from the original on 13 January 2017. Retrieved 13 January 2017.
- ↑ Belluz, Julia. "A woman died from a superbug that outsmarted all 26 US antibiotics". Vox. Archived from the original on 14 January 2017. Retrieved 13 January 2017.
- ↑ "Superbug Killed Nevada Woman". Yahoo! News. Archived from the original on 13 January 2017. Retrieved 13 January 2017.
- ↑ "Guidance : Infection Prevention and Control Measures for Healthcare Workers in All Healthcare Settings" (PDF). Phac-aspc.gc.ca. Archived (PDF) from the original on 25 December 2013. Retrieved 25 October 2017.
- ↑ Yasin, Faizan; Assad, Salman; Talpur, Abdul Subhan; Zahid, Mehr; Malik, Shuja A (2017). "Combination Therapy for Multidrug-Resistant Klebsiella Pneumoniae Urinary Tract Infection". Cureus. 9 (7): e1503. doi:10.7759/cureus.1503. ISSN 2168-8184. PMC 5608481. PMID 28948123.
- ↑ Wilson WC, Grande CM, Hoyt DB (2007). Trauma critical care. New York: Informa Healthcare. p. 444. ISBN 978-1-4200-1684-0. Archived from the original on 11 December 2021. Retrieved 13 January 2017.
- ↑ Sanchez GV, Master RN, Clark RB, Fyyaz M, Duvvuri P, Ekta G, Bordon J (January 2013). "Klebsiella pneumoniae antimicrobial drug resistance, United States, 1998–2010". Emerging Infectious Diseases. 19 (1): 133–6. doi:10.3201/eid1901.120310. PMC 3557979. PMID 23260464.
- ↑ Antoniadou A, Kontopidou F, Poulakou G, Koratzanis E, Galani I, Papadomichelakis E, Kopterides P, Souli M, Armaganidis A, Giamarellou H (April 2007). "Colistin-resistant isolates of Klebsiella pneumoniae emerging in intensive care unit patients: first report of a multiclonal cluster". The Journal of Antimicrobial Chemotherapy. 59 (4): 786–90. doi:10.1093/jac/dkl562. PMID 17307769.
- ↑ "Klebsiella pneumoniae in Healthcare Settings". Centers for Disease Control and Prevention. Archived from the original on 19 October 2021. Retrieved 13 January 2017.
- ↑ Kumarasamy, Karthikeyan K; Toleman, Mark A; Walsh, Timothy R; Bagaria, Jay; Butt, Fafhana; Balakrishnan, Ravikumar; Chaudhary, Uma; Doumith, Michel; Giske, Christian G; Irfan, Seema; Krishnan, Padma; Kumar, Anil V; Maharjan, Sunil; Mushtaq, Shazad; Noorie, Tabassum; Paterson, David L; Pearson, Andrew; Perry, Claire; Pike, Rachel; Rao, Bhargavi; Ray, Ujjwayini; Sarma, Jayanta B; Sharma, Madhu; Sheridan, Elizabeth; Thirunarayan, Mandayam A; Turton, Jane; Upadhyay, Supriya; Warner, Marina; Welfare, William; Livermore, David M; Woodford, Neil (2010). "Emergence of a new antibiotic resistance mechanism in India, Pakistan, and the UK: a molecular, biological, and epidemiological study". The Lancet. Infectious Diseases. 10 (9): 597–602. doi:10.1016/S1473-3099(10)70143-2. ISSN 1473-3099. Archived from the original on 23 August 2022. Retrieved 6 October 2022.
- ↑ Shakil, S.; Azhar, E. I.; Tabrez, S.; Kamal, M. A.; Jabir, N. R.; Abuzenadah, A. M.; Damanhouri, G. A.; Alam, Q. (October 2011). "New Delhi metallo-β-lactamase (NDM-1): an update". Journal of Chemotherapy (Florence, Italy). 23 (5): 263–265. doi:10.1179/joc.2011.23.5.263. ISSN 1973-9478. Archived from the original on 15 May 2021. Retrieved 9 October 2022.
- ↑ Wang, J. H.; Liu, Y. C.; Lee, S. S.; Yen, M. Y.; Chen, Y. S.; Wang, J. H.; Wann, S. R.; Lin, H. H. (June 1998). "Primary liver abscess due to Klebsiella pneumoniae in Taiwan". Clinical Infectious Diseases: An Official Publication of the Infectious Diseases Society of America. 26 (6): 1434–1438. doi:10.1086/516369. ISSN 1058-4838. Archived from the original on 15 June 2022. Retrieved 11 October 2022.
- ↑ 45.0 45.1 Chang, De; Sharma, Lokesh; Dela Cruz, Charles S.; Zhang, Dong (2021). "Clinical Epidemiology, Risk Factors, and Control Strategies of Klebsiella pneumoniae Infection". Frontiers in Microbiology. 12. ISSN 1664-302X. Archived from the original on 20 June 2022. Retrieved 9 October 2022.
- ↑ 46.0 46.1 Clegg, Steven; Murphy, Caitlin N. (19 April 2016). "Epidemiology and Virulence of Klebsiella pneumoniae". Urinary Tract Infections. ASM Press. pp. 435–457. ISBN 978-1-68367-065-0. Retrieved 10 October 2022.
- ↑ Köhler, W.; Mochmann, H. (1988). "[Edwin Klebs (1834-1913), pathologist and forerunner of bacteriology. In memory of the 75th anniversary of his death]". Zeitschrift Fur Arztliche Fortbildung. 82 (20): 1037–1042. ISSN 0044-2178. Archived from the original on 6 October 2022. Retrieved 6 October 2022.
- ↑ "Carl Friedländer (1847–87)". Nature. 160 (4072): 668–668. 1 November 1947. doi:10.1038/160668b0. ISSN 1476-4687. Archived from the original on 16 October 2022. Retrieved 13 October 2022.
- ↑ Garrison, F.H. (1913). "Edwin Klebs (1834-1913)". Science. 38 (991): 920–921. Bibcode:1913Sci....38..920G. doi:10.1126/science.38.991.920. JSTOR 1639550. PMID 17753538. Archived from the original on 2020-11-16. Retrieved 2022-10-14.
- ↑ Anonymous (1913). "Theodor Albrecht Edwin Klebs. Born Königsberg-i.-Pr., February 6, 1834-died Berne, October 23, 1913". The Journal of Pathology and Bacteriology. 18 (1): 401–403. doi:10.1002/path.1700180140.
- ↑ Zander DS, Farver CF (2016). Pulmonary Pathology: A Volume in Foundations in Diagnostic Pathology Series. Elsevier Health Sciences. p. 169. ISBN 978-0-323-46119-1. Archived from the original on 11 December 2021. Retrieved 14 January 2017.
- ↑ Rashid, T; Ebringer, A (June 2007). "Ankylosing spondylitis is linked to Klebsiella – the evidence". Clinical Rheumatology. 26 (3): 858–864. doi:10.1007/s10067-006-0488-7. PMID 17186116. S2CID 43456525.
- ↑ "Archive copy" (PDF). Archived from the original (PDF) on 2020-10-15. Retrieved 2021-10-09.
{{cite web}}: CS1 maint: archived copy as title (link) - ↑ Kaushik, Purshotam; Chauhan, Abhishek (8 March 2009). Cyanobacteria: Antibacterial Activity. New India Publishing. p. 77. ISBN 978-81-907237-7-0. Archived from the original on 18 October 2022. Retrieved 18 October 2022.
- ↑ Bogovazova, GG; Voroshilova, NN; Bondarenko, VM (April 1991). "The efficacy of Klebsiella pneumoniae bacteriophage in the therapy of experimental Klebsiella infection". Zhurnal Mikrobiologii, Epidemiologii, I Immunobiologii (in русский) (4): 5–8. ISSN 0372-9311. PMID 1882608.
- ↑ Chanishvili, N, ed. (2012). A Literature Review of the Practical Application of Bacteriophage Research. Hauppauge, NY: Nova Science. ISBN 978-1-62100-851-4.
- ↑ "Taxonomy browser (Klebsiella)". www.ncbi.nlm.nih.gov. Archived from the original on 20 September 2022. Retrieved 4 October 2022.
- ↑ "Dictyostelium Growth". dictybase.org. Archived from the original on 4 October 2022. Retrieved 4 October 2022.
External links
| Wikispecies has information related to Klebsiella pneumoniae |
- Virtual museum of bacteria page on K. pneumoniae Archived 2017-02-11 at the Wayback Machine
- What're the complications of pneumonia? (health-cares.net)
- Klebsiella Infection Archived 2008-11-06 at the Wayback Machine (emedicine.com)
- Klebsiella Genome Projects Archived 2007-09-27 at the Wayback Machine from Genomes OnLine Database Archived 2011-10-15 at the Wayback Machine
- Klebsiella pneumoniae-Associated Vertebral Osteomyelitis After Laparoscopic Cholecystectomy Archived 2019-09-11 at the Wayback Machine
- Type strain of Klebsiella pneumoniae at BacDive – the Bacterial Diversity Metadatabase Archived 2016-09-17 at the Wayback Machine
- Pages with script errors
- Wikipedia articles incorporating the PD-notice template
- CS1 italiano-language sources (it)
- CS1 maint: archived copy as title
- CS1 русский-language sources (ru)
- Articles with 'species' microformats
- Webarchive template wayback links
- Portal templates with all redlinked portals
- Enterobacteriaceae
- Gram-negative bacteria
- Pathogenic bacteria
- Polysaccharide encapsulated bacteria
- Bacteria described in 1886