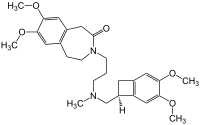
Ivabradine
 | |
 | |
| Names | |
|---|---|
| Pronunciation | /ɪˈvæbrədiːn/ |
| Trade names | Corlanor, Procoralan |
| Other names | Ivabradine hydrochloride, S-16257 |
| |
| Clinical data | |
| Main uses | Heart-related chest pain, heart failure[1] |
| Side effects | Seeing temporary brightness, slow heart rate[1] |
| Routes of use | By mouth |
| Typical dose | 5 mg BID[1] |
| External links | |
| AHFS/Drugs.com | Monograph |
| Legal | |
| License data |
|
| Legal status | |
| Pharmacokinetics | |
| Bioavailability | 40% |
| Protein binding | 70% |
| Metabolism | Liver (first-pass) >50%, CYP3A4-mediated |
| Elimination half-life | 2 hours |
| Excretion | Kidney and fecal |
| Chemical and physical data | |
| Formula | C27H36N2O5 |
| Molar mass | 468.594 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Ivabradine, sold under the brand name Procoralan among others, is a medication used for long term stable heart-related chest pain and heart failure.[1] It may be used when the heart rate is at least 70 beats per minute and the condition is not fully managed by beta blockers.[1][2] It is taken by mouth.[3]
Common side effects include temporarily seeing brightness and slow heart rate.[1] Other side effects may include atrial fibrillation and angioedema.[3] Use during pregnancy may harm the baby.[3] It works by blocking If current in the sinus node which lowers the heart rate.[1]
Ivabradine was approved for medical use in Europe in 2005 and the United States in 2015.[1][3] It is available as a generic medication.[4][5] In the United Kingdom 4 weeks of medication costs the NHS about £5.[4] In the United States this amount costs about 450 USD.[6]
Medical uses
It is used for the symptomatic treatment of chronic stable angina pectoris in people with normal sinus rhythm who cannot take beta blockers. It is also being used off-label in the treatment of inappropriate sinus tachycardia (IST).[7]
Chest pain
It may be as effective as the beta blocker atenolol[8] and comparable with amlodipine[9] in the management of chronic stable angina.
Heart failure
It is used in combination with beta blockers in people with heart failure with LVEF lower than 35 percent inadequately controlled by beta blockers alone and whose heart rate exceeds 70 beats per minute.[10] In people not sufficiently managed with beta blockers for their heart failure adding ivabradine decreases the risk of hospitalization for heart failure.[2]
Tachycardia
It can be used as a bradycardic agent as it has shown to inhibit the If current.[11] Ivabradine appears to be a good on rate control and not much else; utility appears limited to runaway atrial fibrillation.
Dosage
It is generally started at a dose of 5 mg twice per day, though a dose of 2.5 mg to 7.5 mg twice per day may be used.[1]
Contraindications
Ivabradine is contraindicated in sick sinus syndrome. It should also not be used concomitantly with potent inhibitors of CYP3A4, including azole antifungals (such as ketoconazole), macrolide antibiotics, nefazodone and the antiretroviral drugs nelfinavir and ritonavir.[12]
Use of ivabradine with verapamil or diltiazem is contraindicated.[13]
Side effects
Overall, 14.5% of people experience luminous phenomena (by patients described as sensations of enhanced brightness in a fully maintained visual field). This is probably due to blockage of Ih ion channels in the retina, which are very similar to cardiac If. These symptoms are mild, transient, and fully reversible. In clinical studies, about 1% of all patients had to discontinue the drug because of these sensations, which occurred on average 40 days after the drug was started.[8]
In a large clinical trial, bradycardia (unusually slow heart rate) occurred in 2% and 5% of patients taking ivabradine at doses of 7.5 and 10 mg respectively (compared to 4.3% in those taking atenolol).[8] 2.6–4.8% reported headaches.[8] Other common adverse drug reactions (1–10% of patients) include first-degree AV block, ventricular extrasystoles, dizziness and/or blurred vision.[14]
Mechanism of action
Ivabradine acts on the If (f is for "funny", so called because it had unusual properties compared with other current systems known at the time of its discovery) ion current, which is highly expressed in the sinoatrial node. If is a mixed Na+–K+ inward current activated by hyperpolarization and modulated by the autonomic nervous system. It is one of the most important ionic currents for regulating pacemaker activity in the sinoatrial (SA) node. Ivabradine selectively inhibits the pacemaker If current in a dose-dependent manner. Blocking this channel reduces cardiac pacemaker activity, selectively slowing the heart rate and allowing more time for blood to flow to the myocardium.[15][16] By inhiting the If channel ,Ivabradine reduces the heart rate and workload on the heart. This is relevant in the usage of the medication to treat angina as well as congestive heart failure. This is in contrast to other commonly used rate-reducing medications, such as beta-blockers and calcium channel blockers, which not only reduce heart rate, but also the cardiac contractility. Given the selective decrease in rate without loss of contractility, ivabradine may prove efficacious for treatment of congestive heart failure with reduced ejection fraction.
Ivabradine binds to HCN4 receptors (Potassium/sodium hyperpolarization-activated cyclic nucleotide-gated channel 4), utilizing Y506, F509 and I510 residues.[17]
Society and culture
Approval
Ivabradine was approved by the European Medicines Agency in 2005, and by the United States Food and Drug Administration in 2015.[18]
Names
It is marketed by Amgen under the trade name Corlanor in the United States,[19] and by Servier in the rest of the world under the trade names Procoralan (worldwide), Coralan (in Hong Kong, Singapore, Australia and some other countries), Corlentor (in Armenia, Spain, Italy and Romania), Lancora (in Canada) and Coraxan (in Russia and Serbia). It is also marketed in India under the brand names Ivabrad, Ivabid, IVAMAC and Bradia. During its development, ivabradine was known as S-16257.
Research
Coronary artery disease
The BEAUTIFUL study randomised over 10917 patients having stable coronary artery disease and left ventricle dysfunction (ejection fraction < 40%). Ivabradine did not show a significant reduction in the primary composite endpoint of cardiovascular death, admission to hospital for acute myocardial infarction, and admission to hospital for new onset or worsening heart failure. However, in a prespecified subgroup of patients with a heart rate of more than 70 bpm, ivabradine significantly reduced the following secondary endpoints:[20]
- Coronary events by 22% (P=0.023)
- Fatal and nonfatal myocardial infarction by 36% (P=0.001)
- Coronary revascularization by 30% (P=0.016).
The SIGNIFY trial randomized 19,102 patients with coronary artery disease and a heart rate greater than 70, but without clinical heart failure to ivabradine or placebo in addition to standard therapy. Ivabradine did not improve outcomes in this patient group.[21]
Heart failure
In the SHIFT study, ivabradine significantly reduced the risk of the primary composite endpoint of hospitalization for worsening heart failure or cardiovascular death by 18% (P<0.0001) compared with placebo on top of optimal therapy.[22] These benefits were observed after 3 months of treatment. SHIFT also showed that administration of ivabradine to heart failure patients significantly reduced the risk of death from heart failure by 26% (P=0.014) and hospitalization for heart failure by 26% (P<0.0001). The improvements in outcomes were observed throughout all prespecified subgroups: female and male, with or without beta-blockers at randomization, patients below and over 65 years of age, with heart failure of ischemic or non-ischemic etiology, NYHA class II or class III, IV, with or without diabetes, and with or without hypertension.[23]
Ivabradine, though indicated for chronic heart failure in people who are clinically stable, is not indicated in acute heart failure where the enhanced heart rate represents cardiac reserve. Use could destabilise these people.
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 "Procoralan". Archived from the original on 10 November 2021. Retrieved 1 December 2021.
- ↑ 2.0 2.1 Yancy, CW; Jessup, M; Bozkurt, B; Butler, J; Casey, DE Jr; Colvin, MM; Drazner, MH; Filippatos, G; Fonarow, GC; Givertz, MM; Hollenberg, SM; Lindenfeld, J; Masoudi, FA; McBride, PE; Peterson, PN; Stevenson, LW; Westlake, C (20 May 2016). "2016 ACC/AHA/HFSA Focused Update on New Pharmacological Therapy for Heart Failure: An Update of the 2013 ACCF/AHA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Failure Society of America". Circulation. 134 (13): e282–e293. doi:10.1161/CIR.0000000000000435. PMID 27208050.
- ↑ 3.0 3.1 3.2 3.3 "Ivabradine Monograph for Professionals". Drugs.com. Archived from the original on 21 January 2021. Retrieved 26 November 2021.
- ↑ 4.0 4.1 BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 227. ISBN 978-0857114105.
- ↑ Research, Center for Drug Evaluation and (10 February 2022). "2021 First Generic Drug Approvals". FDA. Archived from the original on 21 June 2022. Retrieved 22 October 2022.
- ↑ "Ivabradine Prices, Coupons & Savings Tips - GoodRx". GoodRx. Retrieved 1 December 2021.
- ↑ Yusuf S, Camm AJ (2003). "Sinus tachyarrhythmias and the specific bradycardic agents: a marriage made in heaven?". J. Cardiovasc. Pharmacol. Ther. 8 (2): 89–105. doi:10.1177/107424840300800202. PMID 12808482. S2CID 25305128.
- ↑ 8.0 8.1 8.2 8.3 Tardif JC, Ford I, Tendera M, Bourassa MG, Fox K (2005). "Efficacy of ivabradine, a new selective I(f) inhibitor, compared with atenolol in patients with chronic stable angina". Eur. Heart J. 26 (23): 2529–36. doi:10.1093/eurheartj/ehi586. PMID 16214830.
- ↑ Ruzyllo W, Tendera M, Ford I, Fox KM (2007). "Antianginal efficacy and safety of ivabradine compared with amlodipine in patients with stable effort angina pectoris: a 3-month randomised, double-blind, multicentre, noninferiority trial". Drugs. 67 (3): 393–405. doi:10.2165/00003495-200767030-00005. PMID 17335297. S2CID 25325838.
- ↑ McMurray, JJ; Adamopoulos, S; Anker, SD; Auricchio, A; Böhm, M; Dickstein, K; Falk, V; Filippatos, G; Fonseca, C; Gomez-Sanchez, MA; Jaarsma, T; Køber, L; Lip, GY; Maggioni, AP; Parkhomenko, A; Pieske, BM; Popescu, BA; Rønnevik, PK; Rutten, FH; Schwitter, J; Seferovic, P; Stepinska, J; Trindade, PT; Voors, AA; Zannad, F; Zeiher, A; ESC Committee for Practice, Guidelines (July 2012). "ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure 2012: The Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2012 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association (HFA) of the ESC". European Heart Journal. 33 (14): 1787–847. doi:10.1093/eurheartj/ehs104. PMID 22611136.
- ↑ V Katsi; G Skalis; M S Kallistratos; K Tsioufis; T Makris; A J Manolis; D Tousoulis (2019). "Ivabradine and Metoprolol in Fixed Dose Combination: When, Why and How to Use It". Pharmacological Research. 146: 104279. doi:10.1016/j.phrs.2019.104279. PMID 31108185.
- ↑ "European Medicine Agency, Procoralan Summary of Product Characteristics" (PDF). Archived (PDF) from the original on 2012-05-19. Retrieved 2010-09-13.
- ↑ "Press release: European Medicines Agency recommends measures to reduce risk of heart problems with Corlentor/Procoralan (ivabradine)". European Medicines Agency. 21 November 2014. Archived from the original on 21 June 2018. Retrieved 14 March 2022.
- ↑ Anonymous (2006). "New medicines: Procoralan". Pharmaceutical Journal. 276 (7386): 131. Archived from the original on 2008-03-17. Retrieved 2021-10-28.
- ↑ Thollon C, Cambarrat C, Vian J, Prost JF, Peglion JL, Vilaine JP (1994). "Electrophysiological effects of S 16257, a novel sino-atrial node modulator, on rabbit and guinea-pig cardiac preparations: comparison with UL-FS 49". Br. J. Pharmacol. 112 (1): 37–42. doi:10.1111/j.1476-5381.1994.tb13025.x. PMC 1910295. PMID 8032660.
- ↑ Sulfi S, Timmis AD (2006). "Ivabradine – the first selective sinus node If channel inhibitor in the treatment of stable angina". Int. J. Clin. Pract. 60 (2): 222–8. doi:10.1111/j.1742-1241.2006.00817.x. PMC 1448693. PMID 16451297.
- ↑ Bucchi, A; Baruscotti, M; Nardini, M; Barbuti, A; Micheloni, S; Bolognesi, M; DiFrancesco, D (2013). "Identification of the molecular site of ivabradine binding to HCN4 channels". PLOS ONE. 8 (1): e53132. Bibcode:2013PLoSO...853132B. doi:10.1371/journal.pone.0053132. PMC 3537762. PMID 23308150.
- ↑ "FDA approves Corlanor to treat heart failure". www.fda.gov. Archived from the original on 2015-04-16. Retrieved 2015-04-16.
- ↑ "Amgen and Servier announce product collaboration". www.servier.com. Archived from the original on 2016-03-04. Retrieved 2015-12-29.
- ↑ Kim Fox; Ian Ford; P Gabriel Steg; Michal Tendera; Prof Roberto Ferrari (2008). "Ivabradine for patients with stable coronary artery disease and left-ventricular systolic dysfunction: a randomised, double-blind, placebo-controlled trial". The Lancet. 372 (9641): 807–816. doi:10.1016/S0140-6736(08)61170-8. PMID 18757088. S2CID 26282333.
- ↑ Fox, Kim; Ford, Ian; Steg, Philippe Gabriel; Tardif, Jean-Claude; Tendera, Michal; Ferrari, Roberto (2014). "Ivabradine in Stable Coronary Artery Disease without Clinical Heart Failure" (PDF). New England Journal of Medicine. 371 (12): 1091–1099. doi:10.1056/NEJMoa1406430. PMID 25176136. Archived (PDF) from the original on 2021-10-31. Retrieved 2021-10-28.
- ↑ Stiles, Steve. "SHIFT: Adding HR-slowing agent ivabradine to HF meds cuts mortality, hospitalization". TheHeart.org. Archived from the original on 10 January 2011. Retrieved 1 April 2011.
- ↑ Swedberg K, Komajda M, Böhm M, Borer JS, Ford I, Dubost-Brama A, Lerebours G, Tavazzi L, on behalf of the SHIFT Investigators (2010). "Ivabradine and outcomes in chronic heart failure (SHIFT): a randomised placebo-controlled study". The Lancet. 376 (9744): 875–885. doi:10.1016/S0140-6736(10)61198-1. PMID 20801500. S2CID 21196422. Archived from the original on 2014-08-29. Retrieved 2021-10-28.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- "Ivabradine hydrochloride". Drug Information Portal. U.S. National Library of Medicine. Archived from the original on 2021-08-05. Retrieved 2021-10-28.
- Pages using duplicate arguments in template calls
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drug has EMA link
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- Articles with changed CASNo identifier
- Antianginals
- Benzocyclobutenes
- Laboratoires Servier
- Phenol ethers
- Benzazepines
- Lactams
- Tertiary amines
- RTT