Istradefylline
 | |
| Names | |
|---|---|
| Trade names | Nourianz, Nouryant[1] |
| Other names | KW-6002 |
| |
| Clinical data | |
| Drug class | Adenosine A2A receptor antagonist[2] |
| Main uses | Parkinson's disease (PD) "off" episodes[3] |
| Side effects | Involuntary muscle movements, constipation, hallucinations, dizziness, nausea, trouble sleeping[3] |
| Routes of use | By mouth |
| Typical dose | 20 mg OD[4] |
| External links | |
| AHFS/Drugs.com | Monograph |
| Legal | |
| License data | |
| Legal status | |
| Pharmacokinetics | |
| Protein binding | 98% |
| Metabolism | Mainly CYP1A1, CYP3A4, and CYP3A5 |
| Elimination half-life | 64–69 hrs |
| Excretion | 68% faeces, 18% urine |
| Chemical and physical data | |
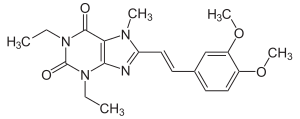
| Formula | C20H24N4O4 |
| Molar mass | 384.436 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Istradefylline, sold under the brand name Nourianz, is a medication used with levodopa/carbidopa for "off" episodes in Parkinson's disease (PD).[3][6] An "off" episode is a time when other medications are not working well, resulting in tremor and difficulty walking.[3] It is taken by mouth.[3]
Common side effects include involuntary muscle movements, constipation, hallucinations, dizziness, nausea, and trouble sleeping.[3] Other side effects may include compulsive behavior.[3] It is a adenosine A2A receptor antagonist.[2]
Istradefylline was approved for medical use in the United States in 2019.[4] In Europe it was refused approval in 2021 due to unclear evidence of benefit.[2] It is not available in the United Kingdom.[1] In the United States it costs about 1,500 USD per month.[7]
Medical uses
Dosage
The typical dose is 20 mg once per day.[4]
Mechanism of action
Istradefylline is a selective antagonist at the adenosine A2A receptor (A2AR), but the precise mechanism by which it exerts its therapeutic effect in Parkinson's disease is unknown.[8] However, it is known that dimers of these receptors form heterotetramers with the dimers of dopamine D2 receptors (D2R) within striatum. Adenosine acts as an endogenous A2AR agonist, but also as a negative allosteric modulator (NAM) within these tetramers towards D2Rs, thus inhibiting the D2R mediated effects of dopamine, an endogenous D2R agonist. Istradefylline is believed to bind a A2AR within a A2AR-D2R-tetramer and function as a NAM towards the other A2AR (instead of D2R), thus inhibiting the effects of adenosine and enhancing the movement (locomotion) promoting effects exerted by dopamine via D2R. However, at high istradefylline concentration, it causes locomotion depression akin to caffeine (which is a broad-spectrum adenosine receptor antagonist), and might do so by displacing adenosine, and working as a NAM towards D2R (instead of A2AR).[9]
History
It was first approved in Japan in 2013.[10]
The effectiveness of Nourianz in treating "off" episodes in patients with Parkinson's disease who are already being treated with levodopa/carbidopa was shown in four 12-week placebo-controlled clinical studies that included a total of 1,143 participants. In all four studies, people treated with Nourianz experienced a statistically significant decrease from baseline in daily "off" time compared to patients receiving a placebo.[11][3]
It was approved for medical use in the United States in 2019.[11][3][12] and approval was granted to Kyowa Kirin, Inc.[11]
The U.S. Food and Drug Administration (FDA) considers it to be a first-in-class medication.[13]
References
- ↑ 1.0 1.1 "Istradefylline". SPS - Specialist Pharmacy Service. 18 September 2019. Archived from the original on 24 May 2021. Retrieved 1 December 2021.
- ↑ 2.0 2.1 2.2 "Nouryant". Archived from the original on 14 November 2021. Retrieved 1 December 2021.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 3.8 "Drug Trials Snapshots: Nourianz". U.S. Food and Drug Administration (FDA). 23 September 2019. Archived from the original on 20 November 2019. Retrieved 19 November 2019.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ 4.0 4.1 4.2 "Istradefylline Monograph for Professionals". Drugs.com. Archived from the original on 18 October 2019. Retrieved 26 November 2021.
- ↑ "Nourianz- istradefylline tablet, film coated". DailyMed. Archived from the original on 2 November 2021. Retrieved 4 January 2021.
- ↑ Cabreira V, Soares-da-Silva P, Massano J (April 2019). "Contemporary Options for the Management of Motor Complications in Parkinson's Disease: Updated Clinical Review". Drugs. 79 (6): 593–608. doi:10.1007/s40265-019-01098-w. PMID 30905034. S2CID 85456263.
- ↑ "Istradefylline Prices, Coupons & Savings Tips - GoodRx". GoodRx. Retrieved 1 December 2021.
- ↑ "Mechanism of Action". Archived from the original on 11 August 2020. Retrieved 27 December 2020.
- ↑ Ferré S, et al. (2018). "New developments on the adenosine mechanisms of the central effects of caffeine and their implications for neuropsychiatric disorders". Journal of Caffeine and Adenosine Research. 8 (4): 121–131. doi:10.1089/caff.2018.0017. PMC 6306650. PMID 30596206.
- ↑ Dungo R, Deeks ED (June 2013). "Istradefylline: first global approval". Drugs. 73 (8): 875–82. doi:10.1007/s40265-013-0066-7. ISSN 1179-1950. PMID 23700273. S2CID 35937325.
- ↑ 11.0 11.1 11.2 "FDA approves new add-on drug to treat off episodes in adults with Parkinson's disease". U.S. Food and Drug Administration (FDA) (Press release). 27 August 2019. Archived from the original on 4 September 2019. Retrieved 29 August 2019.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ "Drug Approval Package: Nourianz". U.S. Food and Drug Administration (FDA). Archived from the original on 2 April 2021. Retrieved 25 October 2021.
- ↑ "New Drug Therapy Approvals 2019". U.S. Food and Drug Administration. 31 December 2019. Archived from the original on 16 September 2020. Retrieved 15 September 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- Pages using duplicate arguments in template calls
- Wikipedia articles incorporating the PD-notice template
- Use dmy dates from October 2019
- Articles with invalid date parameter in template
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Adenosine receptor antagonists
- Alkene derivatives
- Phenol ethers
- Xanthines
- RTT