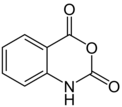
Isatoic anhydride
| |
| Names | |
|---|---|
| Preferred IUPAC name
2H-3,1-Benzoxazine-2,4(1H)-dione | |
| Identifiers | |
| ECHA InfoCard | 100.003.869 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| Properties | |
| C8H5NO3 | |
| Molar mass | 163.132 g·mol−1 |
| Appearance | white solid |
| Melting point | 243 °C (469 °F; 516 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Isatoic anhydride is an organic compound derived from anthranilic acid. A white solid, it is prepared by reaction of anthranilic acid with phosgene.[1]
Reactions
Hydrolysis gives carbon dioxide and anthranilic acid. Alcoholysis proceeds similarly, affording the ester:
- C6H4C2O3NH + ROH → C6H4(CO2R)(NH2) + CO2
Amines also effect ring-opening. Active methylene compounds and carbanions replace oxygen giving hydroxyquinolinone derivatives. Deprotonation followed by alkylation gives the N-substituted derivatives. Sodium azide gives the benzimidazolone via the isocyanate.[2] Isatoic anhydride is used as a blowing agent in the polymer industry, an application that exploits its tendency to release CO2.
Uses
Isatoic anhydride has been used as a precursor for the synthesis of methaqualone and related 4-quinazolinone-based pharmaceutical drugs, including:[3]
References
- ^ E. C. Wagner; Marion F. Fegley. (1947). "Isatoic anhydride". Org. Synth. 27: 45. doi:10.15227/orgsyn.027.0045.
- ^ Coppola, Gary M. (1980). "The Chemistry of Isatoic Anhydride". Synthesis. 7 (7): 505–36. doi:10.1055/s-1980-29110.
- ^ Etienne F. van Zyl (2001). "A survey of reported syntheses of methaqualone and some positional and structural isomers". Forens. Sci. Int. 122: 142–149.
- Articles without InChI source
- Chemical pages without ChemSpiderID
- Articles without EBI source
- Articles without KEGG source
- ECHA InfoCard ID from Wikidata
- Articles containing unverified chemical infoboxes
- Chembox image size set
- Articles with short description
- Short description matches Wikidata
- Carboxylic anhydrides