Iodinated contrast
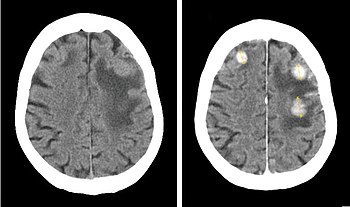
Iodinated contrast is a form of intravenous radiocontrast agent containing iodine, which enhances the visibility of vascular structures and organs during radiographic procedures. Some pathologies, such as cancer, have particularly improved visibility with iodinated contrast.
The radiodensity of iodinated contrast is 25–30 Hounsfield units (HU) per milligram of iodine per milliliter at a tube voltage of 100–120 kVp.[1]
Types
Iodine-based contrast media are usually classified as ionic or nonionic. Both types are used most commonly in radiology due to their relatively harmless interaction with the body and its solubility. Contrast media are primarily used to visualize vessels and changes in tissues on radiography and CT (computerized tomography). Contrast media can also be used for tests of the urinary tract, uterus and fallopian tubes. It may cause the patient to feel as if they have had urinary incontinence. It also puts a metallic taste in the mouth of the patient.
The iodine may be bound either in an organic (nonionic) compound or an ionic compound. Ionic agents were developed first and are still in widespread use depending on the requirements but may result in additional complications due to the high concentration of ions (hyperosmolality). Organic agents which covalently bind the iodine have fewer side effects as they do not dissociate into component molecules. Many of the side effects are due to the hyperosmolar solution being injected. i.e. they deliver more iodine atoms per molecule. The more iodine, the more "dense" the X-ray effect.
Organic iodine molecules used for contrast include iohexol, iodixanol, and ioversol. Iodine-based contrast media used nowadays are water-soluble. These contrast agents are sold as clear, colorless water solutions, with the concentration usually expressed as mg I/ml. Modern iodinated contrast agents can be used almost anywhere in the body. Most often they are used intravenously, but for various purposes they can also be used intra-arterially, intrathecally (as in diskography of the spine) and intra-abdominally – just about any body cavity or potential space.
Heating non-ionic contrast agent to human body temperature reduces its viscosity.[2]
Iodine contrast agents are used for the following:
- Contrast CTs
- Angiography (arterial investigations)
- Venography (venous investigations)
- VCUG (voiding cystourethrography)
- HSG (hysterosalpingogram)
- IVU (intravenous urography)
| Name | Type | Iodine content | Osmolality | |
|---|---|---|---|---|
| diatrizoate (Hypaque 50/ Gastrografin) | Monomer | 300 mgI/ml | 1550 | High |
| metrizoate (Isopaque 370) | Monomer | 370 mgI/ml | 2100 | High |
| iothalamate (Conray) | 600-2400 | High | ||
| ioxaglate (Hexabrix) | Dimer | 320 mgI/ml | 580 | Low |
| Name | Type | Iodine content | Osmolality | |
|---|---|---|---|---|
| iopamidol (Isovue 370) | Monomer | 370 mgI/ml | 796 | Low |
| iohexol (Omnipaque 350) | Monomer | 350 mgI/ml | 884 | Low |
| ioxilan (Oxilan 350) | Monomer | 350 mgI/ml | 695 | Low |
| iopromide (Ultravist 370) | Monomer | 370 mgI/ml | 774 | Low |
| iodixanol (Visipaque 320) | Dimer | 320 mgI/ml | 290 | Low |
| iobitridol (Xenetix 300) | Monomer | 300 mgI/ml | 695 | Low |
| ioversol | ||||
Adverse effects
Iodinated contrast agents may cause allergic reactions, contrast-induced nephropathy, hyperthyroidism and possibly metformin accumulation. However, there are no absolute contraindications to iodinated contrast, so the benefits needs to be weighted against the risks.[3]
In people with myasthenia gravis, older forms of iodinated contrast have caused an increased risk of exacerbation of the disease, but modern forms have no immediate increased risk.[4]
Hypersensitivity reactions
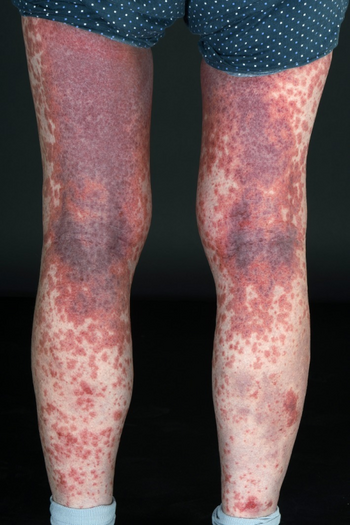
Anaphylactoid reactions occur rarely,[5][6][7] but can occur in response to injected as well as oral and rectal contrast and even retrograde pyelography. They are similar in presentation to anaphylactic reactions, but are not caused by an IgE-mediated immune response. Patients with a history of contrast reactions, however, are at increased risk of anaphylactoid reactions.[8][9] Pretreatment with corticosteroids has been shown to decrease the incidence of adverse reactions.[10][11]
Anaphylactoid reactions range from urticaria and itching, to bronchospasm and facial and laryngeal edema. For simple cases of urticaria and itching, an oral or intravenous antihistamine such as diphenhydramine is appropriate. For more severe reactions, including bronchospasm and facial or neck edema, albuterol inhaler, or subcutaneous or IV epinephrine, plus diphenhydramine may be needed. If respiration is compromised, an airway must be established prior to medical management.
Anaphylaxis to ionic (high osmolar) contrast agent injections occurred in two clusters of reactions on two occasions (1983 and 1987) in a single radiology clinic in London, Ontario. On each occasion, these anaphylactic reactions were associated with contamination of the injection by natural rubber components (disposable plastic syringes in the first case and rubber ampoule seals in the second case). The allergenic-toxic rubber leachate was MBT (mercaptobenzothiazole). This is a known allergen that becomes bound to plasma proteins, creating a hapten-protein complex – a signature mechanism in true IgE drug allergy and true anaphylactic reactions (not "anaphylactoid" reactions).
A Japanese syringe manufacturer, Terumo, implicated in syringe-related toxic laboratory cell culture effects in Australia in 1981, was instrumental in pro-actively making Japanese disposable syringes and ampoule seals free of natural rubber. Katayama's 1990 article in Radiology showed that a new type of nonionic (low osmolar) contrast agent was associated with significantly fewer severe life-threatening reactions than the older ionic (high osmolar) contrast agents.[12] By merchandizing the Katayama series reprints, manufacturers persuaded users worldwide to switch to the almost exclusive use of the expensive nonionic agents.
What was unknown to the Katayama researchers was that the ampoule seals of the "safer" nonionic contrast agents were made from artificial rubber, whereas the ionic agents were sealed with natural rubber. In 1987, it was the leaching of allergenic MBT from the rubber seals of ionic ampoules that caused a series of allergic reactions (including anaphylaxis) in a radiology office in Canada.[13] The worldwide hazard of MBT contamination of injections was unknown then and, as the World Health Organization reported it remains as an unknown hazard still – after three decades.[14]
The most significant study, proving that injections of ionic (high osmolar) agents are at least as safe as the newer, very expensive nonionic agents was published in Radiology in 1997.[15] Lasser did not comment that the marked drop in the incidence of severe reactions with ionic agents was related to the removal of natural rubber contamination from ionic ampoule seals.
Contribution of seafood and other allergies
The term "iodine allergy" should be omitted because this kind of allergy does not exist.[16] Seafood "allergy" is not a contraindication for the use of iodinated contrast materials, because in seafood allergy the immune system is directed against the muscle protein tropomyosin. While iodine levels in seafood are higher than in non-seafood items, the consumption of the latter exceeds that of the former by far and there is no evidence that the iodine content of seafood is related to reactions to seafood.[17] Available data suggest that seafood allergy increases the risk of a contrast-mediated reaction by approximately the same amount as allergies to fruits or those with asthma. In addition, those with an intolerance to alcohol should avoid use of this product due to chemical breakdown similar to ethyl alcohol. Studies show that B.A.C. has been shown to increase exponentially for up to 72 hours after contrast is administered, resulting in altered results of urine, blood, and breath alcohol screens.[18] Over 85% of patients with seafood allergies will not have an adverse reaction to iodinated contrast.[17] Finally, there is no evidence that adverse skin reactions to iodine-containing topical antiseptics (e.g., povidone-iodine) are of any specific relevance to administration of I.V. contrast material.[17][19]
IL-2 medication poses no risk for the acquisition of adverse events by radiocontrast agents.[20]
Contrast-induced nephropathy
Contrast-induced nephropathy is defined as either a greater than 25% increase of serum creatinine or an absolute increase in serum creatinine of 0.5 mg/dL.[21] Iodinated contrast may be toxic to the kidneys, especially when given via the arteries prior to studies such as catheter coronary angiography. Nonionic contrast agents, which are almost exclusively used in CT scans, have not been shown to cause CIN when given intravenously at doses needed for CT studies.[22]
Effects on thyroid function
Iodinated contrast media exposure can potentially cause incident hyperthyroidism and incident overt hypothyroidism."[23][24] Hyperthyroidism is the effect of iodine being a substrate of thyroid hormones, and is then called the Jod-Basedow phenomenon. The risk is higher in those with an underlying thyroid disease, such as toxic multinodular goiter, Graves’ disease, or Hashimoto’s thyroiditis, where thyroid monitoring is indicated.[25] Otherwise, for the general population, routine screening with thyroid function tests is generally not feasible.[25]
Drug interactions
It has been recommended that metformin, an oral antidiabetic agent, be stopped for 48 hours following the intravascular administration of contrast media and that the use of metformin not be resumed until kidney function has been shown to be normal. The reasoning is that if the contrast medium causes kidney failure (as happens rarely) and the person continues to take metformin (which is normally excreted by the kidneys), there may be a toxic accumulation of metformin, increasing the risk of lactic acidosis, a dangerous complication.[26]
However, guidelines published by the American College of Radiologists suggest this is not as important for patients who have normal kidney function and no evidence of acute kidney injury. If kidney impairment is found before administration of the contrast, metformin should be withheld for 48 hours following the procedure and until kidney function has returned to normal.[27]
Contrast exposure may interfere with subsequent radioiodine treatment, causing unwanted delays in the management of thyroid cancer.[25]
Previously, beta blockers have been assumed as risk factor for the acquisition of contrast medium-induced adverse reactions/hypersensitivity reactions. Due to recent investigations it became clear that beta blockers do not increase the frequency of adverse reactions in concert with radiocontrast agents.[28]
Pregnancy
Iodinated contrast in medical imaging in pregnancy, when orally administered, is harmless.[29] Intravenous administration of iodinated radiocontrast agents can cross the placenta and enter the fetal circulation, but animal studies have reported no teratogenic or mutagenic effects from its use. There have been theoretical concerns about potential harm of free iodide on the fetal thyroid gland,[29] but multiple studies have shown that a single dose of intravenously administered iodinated contrast medium to a pregnant mother has no effect on neonatal thyroid function.[30] Nevertheless, it generally is recommended that radiocontrast only be used if absolutely required to obtain additional diagnostic information that will improve the care of the fetus or mother.[29]
Breastfeeding
American College of Radiology (ACR) guidelines state that iodinated contrast administration to a breastfeeding mother is considered safe for both the mother and child.[31] Still, mothers who remain concerned about any potential adverse effects to the child are recommended to have the option of abstaining from breastfeeding for 24 hours, with continued milk extraction such as by a breast pump during that period.[31] Mothers that opt for this for non-emergent exams may also use a breast pump to obtain milk before the exam in order to feed the child during the 24-hour abstinence period.[31]
See also
References
- ↑ Bae, Kyongtae T. (2010). "Intravenous Contrast Medium Administration and Scan Timing at CT: Considerations and Approaches". Radiology. 256 (1): 32–61. doi:10.1148/radiol.10090908. ISSN 0033-8419. PMID 20574084.
- ↑ Zhang, Bin; Liu, Jing; Dong, Yuhao; Guo, Baoliang; Lian, Zhouyang; Yu, Hui; Luo, Xiaoning; Mo, Xiaokai; Zhang, Lu; Huang, Wenhui; Ouyang, Fusheng (2018-11-01). "Extrinsic warming of low-osmolality iodinated contrast media to 37°C reduced the rate of allergic-like reaction". Allergy and Asthma Proceedings. 39 (6): e55–e63. doi:10.2500/aap.2018.39.4160. PMC 6212634. PMID 30401329. Archived from the original on 2022-02-07. Retrieved 2022-02-02.
- ↑ Stacy Goergen (2016-09-13). "Iodine-containing contrast medium". InsideRadiology - The Royal Australian and New Zealand College of Radiologists. Archived from the original on 2021-03-03. Retrieved 2019-02-22. Page last modified on 26/7/2017
- ↑ Mehrizi, Mehyar; Pascuzzi, Robert M. (2014). "Complications of radiologic contrast in patients with myasthenia gravis". Muscle & Nerve. 50 (3): 443–444. doi:10.1002/mus.24254. ISSN 0148-639X. PMID 24677227. S2CID 206295540.
- ↑ Karnegis JN, Heinz J (1979). "The risk of diagnostic cardiovascular catheterization". Am Heart J. 97 (3): 291–7. doi:10.1016/0002-8703(79)90427-7. PMID 420067.
- ↑ Lasser EC, Berry CC, Talner LB, Santini LC, Lang EK, Gerber FH, Stolberg HO (1987). "Pretreatment with corticosteroids to alleviate reactions to intravenous contrast material". N Engl J Med. 317 (14): 845–9. doi:10.1056/NEJM198710013171401. PMID 3627208.
- ↑ Greenberger PA, Patterson R, Tapio CM (1985). "Prophylaxis against repeated radiocontrast media reactions in 857 cases. Adverse experience with cimetidine and safety of beta-adrenergic antagonists". Arch Intern Med. 145 (12): 2197–200. doi:10.1001/archinte.145.12.2197. PMID 2866755.
- ↑ Greenberger PA, Patterson R (1988). "Adverse reactions to radiocontrast media". Prog Cardiovasc Dis. 31 (3): 239–48. doi:10.1016/0033-0620(88)90017-5. PMID 3055068.
- ↑ Lang DM, Alpern MB, Visintainer PF, Smith ST (1993). "Elevated risk of anaphylactoid reaction from radiographic contrast media is associated with both beta-blocker exposure and cardiovascular disorders". Arch Intern Med. 153 (17): 2033–40. doi:10.1001/archinte.153.17.2033. PMID 8102844.
- ↑ Lasser EC, Berry CC, Talner LB, Santini LC, Lang EK, Gerber FH, Stolberg HO (1988). "Protective effects of corticosteroids in contrast material anaphylaxis". Invest Radiol. 23 Suppl 1: S193–4. doi:10.1097/00004424-198809001-00035. PMID 3058630.
- ↑ Wittbrodt ET, Spinler SA (1994). "Prevention of anaphylactoid reactions in high-risk patients receiving radiographic contrast media". Ann Pharmacother. 28 (2): 236–41. doi:10.1177/106002809402800215. PMID 8173143. S2CID 25654677.
- ↑ Katayama H; Yamaguchi K.; Kozuka T.; et al. (1990). "Adverse Reactions to Ionic and Nonionic Contrast Media. A Report from the Japanese Committee on the Safety of Contrast Media". Radiology. 175 (3): 621–28. doi:10.1148/radiology.175.3.2343107. PMID 2343107.
- ↑ Hamilton Gavin (1990). "Medical Rubber Anaphylaxis". Lancet. 336 (8728): 1453–1454. doi:10.1016/0140-6736(90)93165-l. PMID 1978917. S2CID 42580307.
- ↑ Book review "The Nurses are Innocent," Lethal, odd and ‘new’ in pharmacovigilance. Uppsala Reports 61 - April 2013: Page 10–11.
- ↑ Lasser E.C.; Lyon G.L.; Berry C.C. (1997). "Reports on Contrast Media Reactions: Analysis of Data from Reports to the U.S. Food and Drug Administration". Radiology. 203 (3): 605–10. doi:10.1148/radiology.203.3.9169676. PMID 9169676.
- ↑ Böhm, I.; Hasembank Keller, P. S.; Heverhagen, J. T. (2016). ""Iodine Allergy" – The Neverending Story" (PDF). Fortschr Röntgenstr. 188 (8): 733–734. doi:10.1055/s-0042-110102. PMID 27459005. Archived (PDF) from the original on 2021-11-04. Retrieved 2022-02-02.
- ↑ 17.0 17.1 17.2 Coakley F, Panicek D (1997). "Iodine allergy: an oyster without a pearl?". AJR Am J Roentgenol. 169 (4): 951–2. doi:10.2214/ajr.169.4.9308442. PMID 9308442.
- ↑ Shehadi W (1975). "Adverse reactions to intravascularly administered contrast media. A comprehensive study based on a prospective survey". Am J Roentgenol Radium Ther Nucl Med. 124 (1): 145–52. doi:10.2214/ajr.124.1.145. PMID 1170768.
- ↑ van Ketel W, van den Berg W (1990). "Sensitization to povidone-iodine". Dermatol Clin. 8 (1): 107–9. doi:10.1016/S0733-8635(18)30531-X. PMID 2302848.
- ↑ Boehm I. Is interleukin-2 (still) a risk factor for adverse reactions in concert with radiographic contrast medium injection? Acta Radiol 2009; 50(7) 752-3
- ↑ Barrett BJ, Parfrey PS (2006). "Clinical practice. Preventing nephropathy induced by contrast medium". N. Engl. J. Med. 354 (4): 379–86. doi:10.1056/NEJMcp050801. PMID 16436769.
- ↑ McDonald, Robert; McDonald, Jennifer S.; Carter, Rickey E.; Hartman, Robert P.; Katzberg, Richard W.; Kallmes, David F.; Williamson, Eric E. (December 2014). "Intravenous Contrast Material Exposure Is Not an Independent Risk Factor for Dialysis or Mortality". Radiology. 273 (3): 714–725. doi:10.1148/radiol.14132418. PMID 25203000.
- ↑ Association Between Iodinated Contrast Media Exposure and Incident Hyperthyroidism and Hypothyroidism Archived 2021-02-10 at the Wayback Machine JAMA
- ↑ O'Connor, Anahad (23 January 2012). "Iodide Heart Scans Linked to Thyroid Disease". New York Times. Archived from the original on 25 January 2012.
- ↑ 25.0 25.1 25.2 Yıldız, Sema; Kuşkonmaz, Şerife Mehlika (2016). "Effect of iodinated contrast media on thyroid: a brief review". Journal of Health Sciences. 6 (1): 12. doi:10.17532/jhsci.2016.278. ISSN 1986-8049.
- ↑ Rasuli P, Hammond DI (1998). "Metformin and contrast media: where is the conflict?". Can Assoc Radiol J. 49 (3): 161–6. PMID 9640281.
- ↑ American College of Radiology (2016). "ACR Manual on Contrast Media" (PDF). p. 46. Archived (PDF) from the original on 1 January 2018. Retrieved 6 January 2017.
- ↑ Boehm I, Morelli J, Nairz K, Silva Hasembank Keller P, Heverhagen JT (2016). "Beta blockers and intravenous roentgen contrast materials; which risks do exist?" (PDF). Eur J Intern Med. 35 (November): e17–e18. doi:10.1016/j.ejim.2016.08.003. PMID 27531627. Archived (PDF) from the original on 2018-07-19. Retrieved 2022-02-02.
- ↑ 29.0 29.1 29.2 "Guidelines for Diagnostic Imaging During Pregnancy and Lactation". American Congress of Obstetricians and Gynecologists. Archived from the original on 2017-07-30. Retrieved 2022-02-02. February 2016
- ↑ "ACR Manual on Contrast Media. Version 10.3" (PDF). American College of Radiology. American College of Radiology Committee on Drugs and Contrast Media. 2017. Archived from the original (PDF) on 2017-09-24. Retrieved 2017-07-30.
- ↑ 31.0 31.1 31.2 "Diagnostic Radiology Procedures on Breastfeeding Patients". Yale School of Medicine. Archived from the original on 2022-02-07. Retrieved 2019-07-08.
Further reading
- Bontranger, Kenneth L. & Lampignano, John P. (2005). Radiographic Positioning and Related Anatomy, St. Louis: Elsevier Mosby. ISBN 0-323-02507-2.