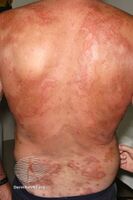
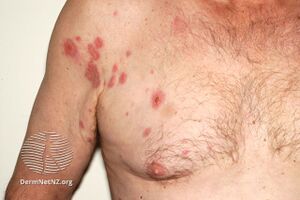
Hypereosinophilic syndrome
| Hypereosinophilic syndrome | |
|---|---|
| Other names: HES[1] | |
 | |
| Hypereosinophilic syndrome | |
| Specialty | Hematology |
| Differential diagnosis | Clonal eosinophilia, Reactive eosinophilia |
Hypereosinophilic syndrome is a disease characterized by a persistently elevated eosinophil count (≥ 1500 eosinophils/mm³) in the blood for at least six months without any recognizable cause, with involvement of either the heart, nervous system, or bone marrow.[2][3]
HES is a diagnosis of exclusion, after clonal eosinophilia (such as FIP1L1-PDGFRA-fusion induced hypereosinophelia and leukemia) and reactive eosinophilia (in response to infection, autoimmune disease, atopy, hypoadrenalism, tropical eosinophilia, or cancer) have been ruled out.[4][5]
There are some associations with chronic eosinophilic leukemia[6] as it shows similar characteristics and genetic defects.[7] If left untreated, HES is progressive and fatal. It is treated with glucocorticoids such as prednisone.[4] The addition of the monoclonal antibody mepolizumab may reduce the dose of glucocorticoids.[8]
Signs and symptoms
As HES affects many organs at the same time, symptoms may be numerous. Some possible symptoms a patient may present with include:
- Cardiomyopathy[6]
- Skin lesions[6]
- Thromboembolic disease[6]
- Pulmonary disease[6]
- Neuropathy[6]
- Hepatosplenomegaly[6]
- Reduced ventricular size[6]
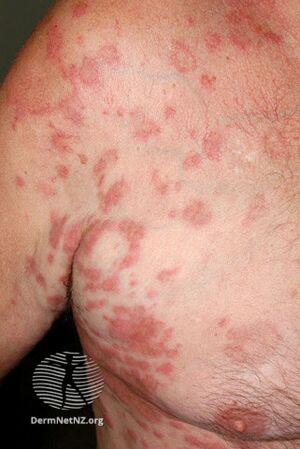
- Atopic eczema
-
Hypereosinophilic syndrome
-
Hypereosinophilic syndrome
Diagnosis
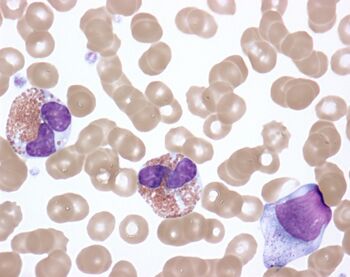
Numerous techniques are used to diagnose hypereosinophilic syndrome, of which the most important is blood testing. In HES, the eosinophil count is greater than 1.5 × 109/L.[7] On some smears the eosinophils may appear normal in appearance, but morphologic abnormalities, such as a lowering of granule numbers and size, can be observed.[7] Roughly 50% of patients with HES also have anaemia.[7]
Secondly, various imaging and diagnostic technological methods are utilised to detect defects to the heart and other organs, such as valvular dysfunction and arrhythmias by usage of echocardiography.[7] Chest radiographs may indicate pleural effusions and/or fibrosis,[7] and neurological tests such as CT scans can show strokes and increased cerebrospinal fluid pressure.[7]
A proportion of patients have a mutation involving the PDGFRA and FIP1L1 genes on the fourth chromosome, leading to a tyrosine kinase fusion protein. Testing for this mutation is now routine practice, as its presence indicates response to imatinib, a tyrosine kinase inhibitor.[9]
Treatment
Treatment primarily consists of reducing eosinophil levels and preventing further damage to organs.[7] Corticosteroids, such as prednisone, are good for reducing eosinophil levels and antineoplastics are useful for slowing eosinophil production.[7] Surgical therapy is rarely utilised, however splenectomy can reduce the pain due to spleen enlargement.[7] If damage to the heart (in particular the valves), then prosthetic valves can replace the current organic ones.[7] Follow-up care is vital for the survival of the patient, as such the patient should be checked for any signs of deterioration regularly.[7] After promising results in drug trials (95% efficiency in reducing blood eosinophil count to acceptable levels) it is hoped that in the future hypereosinophilic syndrome, and diseases related to eosinophils such as asthma and eosinophilic granulomatosis with polyangiitis, may be treated with the monoclonal antibody mepolizumab currently being developed to treat the disease.[8] If this becomes successful, it may be possible for corticosteroids to be eradicated and thus reduce the amount of side effects encountered.[8]
Epidemiology
The European Medicines Agency (EMA) estimated the prevalence of HES at the time of granting orphan drug designation for HES in 2004 at 1.5 in 100,000 people, corresponding to a current prevalence of about 8,000 in the EU, 5,000 in the U.S., and 2,000 in Japan.[10]
Patients who lack chronic heart failure and those who respond well to prednisone or a similar drug have a good prognosis.[7] However, the mortality rate rises in patients with anaemia, chromosomal abnormalities or a very high white blood cell count.[7]
History
Hypereosinophilic syndrome was first described as a distinct entity by Hardy and Anderson in 1968.[11]
References
- ↑ "OMIM Entry - # 607685 - HYPEREOSINOPHILIC SYNDROME, IDIOPATHIC; HES". omim.org. Archived from the original on 12 May 2019. Retrieved 12 May 2019.
- ↑ Chusid MJ, Dale DC, West BC, Wolff SM (1975). "The hypereosinophilic syndrome: analysis of fourteen cases with review of the literature". Medicine (Baltimore). 54 (1): 1–27. doi:10.1097/00005792-197501000-00001. PMID 1090795. S2CID 39212252.
- ↑ Bolognia, Jean L.; Jorizzo, Joseph L.; Rapini, Ronald P. Dermatology. Gulf Professional Publishing. p. 435. ISBN 978-99976-38-99-1. Archived from the original on 2022-11-12. Retrieved 2022-11-05.
- ↑ 4.0 4.1 Fazel R, Dhaliwal G, Saint S, Nallamothu BK (May 2009). "Clinical problem-solving. A red flag". N. Engl. J. Med. 360 (19): 2005–10. doi:10.1056/NEJMcps0802754. PMID 19420370.
- ↑ Reiter A, Gotlib J (2017). "Myeloid neoplasms with eosinophilia". Blood. 129 (6): 704–714. doi:10.1182/blood-2016-10-695973. PMID 28028030.
- ↑ 6.0 6.1 6.2 6.3 6.4 6.5 6.6 6.7 Longmore, Murray; Ian Wilkinson; Tom Turmezei; Chee Kay Cheung (2007). Oxford Handbook of Clinical Medicine. Oxford. p. 316. ISBN 978-0-19-856837-7.
- ↑ 7.00 7.01 7.02 7.03 7.04 7.05 7.06 7.07 7.08 7.09 7.10 7.11 7.12 7.13 Rothenberg, Marc E (2008). "Treatment of Patients with the Hypereosinophilic Syndrome with Mepolizumab". The New England Journal of Medicine. 358 (12): 1215–28. doi:10.1056/NEJMoa070812. PMID 18344568. Archived from the original on 2008-03-12. Retrieved 2008-03-17. Last updated: Updated: Oct 4, 2009 by Venkata Samavedi and Emmanuel C Besa
- ↑ 8.0 8.1 8.2 Rothenberg ME, Klion AD, Roufosse FE, et al. (March 2008). "Treatment of patients with the hypereosinophilic syndrome with mepolizumab". N. Engl. J. Med. 358 (12): 1215–28. doi:10.1056/NEJMoa070812. PMID 18344568. Archived from the original on 2021-08-28. Retrieved 2019-07-08.
- ↑ Cools J, DeAngelo DJ, Gotlib J, et al. (2003). "A tyrosine kinase created by fusion of the PDGFRA and FIP1L1 genes as a therapeutic target of imatinib in idiopathic hypereosinophilic syndrome". N. Engl. J. Med. 348 (13): 1201–14. doi:10.1056/NEJMoa025217. PMID 12660384.
- ↑ European Medicines Agency, Committee for Orphan Medicinal Products. Public summary of opinion on orphan designation: mepolizumab for the treatment of hypereosinophilic syndrome. August 2010.
- ↑ Hardy, William R.; Anderson, Robert E. (1 June 1968). "The Hypereosinophilic syndromes". Annals of Internal Medicine. 68 (6): 1220–9. doi:10.7326/0003-4819-68-6-1220. eISSN 1539-3704. ISSN 0003-4819. PMID 5653621.
External links
| Classification | |
|---|---|
| External resources |