Formoterol
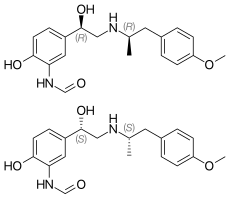 | |
 Formoterol (top), (R,R)-(−)-formoterol (center) and (S,S)-(+)-formoterol (bottom) | |
| Names | |
|---|---|
| Trade names | Oxeze, Perforomist, others |
| |
| Clinical data | |
| Drug class | LABA[1] |
| Main uses | Asthma, COPD[2] |
| Side effects | Dry mouth, trouble sleeping, nausea, throat inflammation[1] |
| Pregnancy category |
|
| Routes of use | Inhalation (capsules for oral inhalation, DPI, MDI) |
| Onset of action | Up to 3 hr for max. effect[1] |
| Duration of action | 12 h[1] |
| External links | |
| AHFS/Drugs.com | Monograph |
| Legal | |
| License data | |
| Legal status | |
| Pharmacokinetics | |
| Protein binding | 61% to 64% |
| Metabolism | Liver demethylation and glucuronidation (CYP2D6, CYP2C19, CYP2C9 and CYP2A6 involved) |
| Elimination half-life | 10 h |
| Excretion | Kidney and fecal |
| Chemical and physical data | |
| Formula | C19H24N2O4 |
| Molar mass | 344.411 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture |
| |
| |
Formoterol, also known as eformoterol, is a used in the long term management of asthma and chronic obstructive pulmonary disease (COPD).[2] It is used by breathing it into the lungs.[2] Full effects take about 2 hours and last about 12 h.[1]
Common side effects include dry mouth, trouble sleeping, nausea, and throat inflammation.[1] Other side effects may include bronchospasm, anaphylaxis, and QT prolongation.[1] In asthma, it may increase the risk of death if used without an inhaled steroid.[1] Use in pregnancy is of unclear safety.[1] It is a long-acting β2 agonist (LABA).[1]
Formoterol was patented in 1972 and came into medical use in 1998.[4] It is available as a generic medication.[5] In the United Kingdom it costs the NHS about £12 per month as of 2021.[2] In the United States this amount costs about 285 USD.[5] It is also available in combination as budesonide/formoterol and mometasone/formoterol.[2][1]
Medical uses
Dosage
It is often used at a dose of 12 micrograms twice per day.[2]
Combinations
- Combination drugs:
- Arformoterol ((R,R)-(−)-formoterol) — an enantiopure compound used in the management of COPD
Side effects
In November 2005, the US Food and Drug Administration (FDA) released a health advisory alerting the public to findings that show the use of long-acting β2 agonists could lead to a worsening of wheezing symptoms in some.[6]
Mechanism of action
Inhaled formoterol works like other β2 agonists, causing bronchodilation by relaxing the smooth muscle in the airway so as to treat the exacerbation of asthma.
Society and culture
Brand names

It is marketed in three forms: a dry-powder inhaler (DPI), a metered-dose inhaler (MDI) and an inhalation solution, under various brand names including Atock, Atimos/Atimos Modulite, Foradil/Foradile, Oxeze/Oxis, and Perforomist.
- Foradil/Foradile capsules for oral inhalation (Schering-Plough in the U.S., Novartis rest of world)
- Oxeze/Oxis Turbuhaler Dry powder inhaler (DPI) (AstraZeneca)
- Atock (Astellas)
- Atimos/Atimos Modulite Metered-dose inhaler (MDI) (Chiesi)
- Perforomist inhalation solution (Mylan N.V.)
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 "Formoterol Monograph for Professionals". Drugs.com. Archived from the original on 28 November 2021. Retrieved 13 December 2021.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 267. ISBN 978-0857114105.
- ↑ "Archive copy" (PDF). Archived (PDF) from the original on 2021-10-31. Retrieved 2021-07-12.
{{cite web}}: CS1 maint: archived copy as title (link) - ↑ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 543. ISBN 9783527607495. Archived from the original on 2021-07-24. Retrieved 2021-07-12.
- ↑ 5.0 5.1 "Formoterol Prices, Coupons & Savings Tips - GoodRx". GoodRx. Retrieved 13 December 2021.
- ↑ "Advair Diskus, Advair HFA, Brovana, Foradil, Perforomist, Serevent Diskus, and Symbicort Information (Long Acting Beta Agonists)". U.S. Food and Drug Administration (FDA). Archived from the original on 2017-11-01. Retrieved 2021-07-12.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- "Formoterol fumarate". Drug Information Portal. U.S. National Library of Medicine. Archived from the original on 2021-04-15. Retrieved 2021-07-12.
- Pages using duplicate arguments in template calls
- CS1 maint: archived copy as title
- Articles with hatnote templates targeting a nonexistent page
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drug has EMA link
- Beta2-adrenergic agonists
- Phenol ethers
- Phenols
- Schering-Plough brands
- Merck & Co. brands
- AstraZeneca brands
- Novartis brands
- Formamides
- Substituted amphetamines
- Phenylethanolamines
- RTT