Fibroadenoma
| Fibroadenoma | |
|---|---|
| Other names: Breast mice, breast mouse[1] | |
 | |
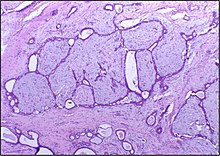
| Histopathologic image of breast fibroadenoma. Core needle biopsy. H&E stain. | |
| Specialty | Gynecology |
| Symptoms | Round, firm, and easy to move breast lump up to 3 cm[2][3] |
| Usual onset | 15 and 35 years[3] |
| Types | Pericanalicular, intracanalicular[4] |
| Causes | Unknown[1] |
| Risk factors | Pregnancy, hormone replacement therapy[2] |
| Diagnostic method | Examination, ultrasound, mammography, needle biopsy[3][1] |
| Differential diagnosis | Breast cancer, phyllodes tumors, lipoma, breast cyst, fat necrosis[3][1] |
| Treatment | None, observation, removal by surgery or biopsy[3][1] |
| Frequency | 25% of women[3] |
Fibroadenomas, also known as breast mice, are a type of non-serious breast tumour.[2] They often feel round, firm, and are easy to move within the breast.[2] They are typically up to 3 cm in size and are not painful.[2][3] In about 70% of cases they occur singularly.[1] They are not associated with breast cancer;[2] though may result in anxiety or unequal breast size.[1]
The cause is unknown.[1] They may become more noticeable during pregnancy or hormone replacement therapy and shrink following menopause.[2] They contain a mixture of connective and glandular tissue.[2] Diagnosis may be supported by examination, ultrasound, mammography, and needle biopsy.[3][1] It involves ruling out other types of breast lumps including breast cancer and phyllodes tumors.[3]
If there are no symptoms, no treatment beyond reassurance and follow-up is required.[3][1] In cases that are bothersome, removal by surgery or biopsy is an option.[3] Other techniques that are being looked at include cryoablation and high-intensity focused ultrasound (HIFU).[3] Fibroadenomas affect about 25% of women.[3] They most often begin between the age of 15 and 35 years.[3] The name breast mice refers to their high mobility within the breast.[1]
Signs and symptoms
Fibroadenomas are non-serious tumours of the breast, most often being seen in women in their 20s and 30s.[5] They are usually solid breast lumps that are:
People who have a simple fibroadenoma do have an increased risk of developing breast cancer compared to the general population.[5] Complex fibroadenomas may increase the risk of breast cancer slightly.[5]
In the male breast, fibroepithelial tumors are very rare, and are mostly phyllodes tumors. Exceptionally rare case reports exist of fibroadenomas in the male breast; however, these cases may be associated with antiandrogen treatment.[6]
Cause
The cause of fibroadenoma is unknown.[1] A connection between fibroadenomas and reproductive hormones has been suggested which may explain why they present themselves during reproductive years, increase in size during pregnancy, and regress post-menopause.[7]
Higher intake of fruits and vegetables, higher number of live births, lower use of oral contraceptives and moderate exercise are associated with lower frequency of fibroadenomas.[8]
Diagnosis
A fibroadenoma is usually diagnosed through clinical examination, ultrasound or mammography, and often a biopsy sample of the lump.[9] Suspicious findings on imaging may result in a person needing a biopsy in order to gain a definitive diagnosis. There are three types of biopsies: fine-needle aspiration, core-needle biopsy and surgical biopsy. The method of biopsy depends on the appearance, size and location of the breast mass.[10]
Pathology


Cytology
The diagnostic findings on needle biopsy consist of abundant stromal cells, which appear as bare bipolar nuclei, throughout the aspirate; sheets of fairly uniform-size epithelial cells that are typically arranged in either an antler-like pattern or a honeycomb pattern. These epithelial sheets tend to show typical metachromatic blue on Diff-Quik staining. Foam cells and apocrine cells may also be seen, although these are less diagnostic features.[9][11] The gallery images below demonstrate these features.
Cellular fibroadenoma, also known as juvenile fibroadenoma, is a variant type of fibroadenoma with increased stromal cellularity.[12][13]
Macroscopic
Approximately 90% of fibroadenomas are less than 3 cm in diameter. However, these tumors have the potential to grow reaching a remarkable size, particularly in young individuals. The tumor is round or ovoid, elastic, and nodular, and has a smooth surface. The cut surface usually appears homogenous and firm, and is grey-white or tan in colour. The pericanalicular type (hard) has a whorly appearance with a complete capsule, while the intracanalicular type (soft) has an incomplete capsule.[11]
Microscopic
Fibroadenoma of the breast is a benign tumor composed of a biplastic proliferation of both stromal and epithelial components.[14][15] This biplasia can be arranged in two growth patterns: pericanalicular (stromal proliferation around epithelial structures) and intracanalicular (stromal proliferation compressing the epithelial structures into slit-like spaces).
These tumors characteristically display hypovascular stroma compared to malignant neoplasms.[16][17][11] Furthermore, the epithelial proliferation appears in a single terminal ductal unit and describes duct-like spaces surrounded by a fibroblastic stroma. The basement membrane is intact.[18]
Molecular pathology
Up to 66% of fibroadenomas harbor mutations in the exon (exon 2) of the mediator complex subunit 12 (MED12) gene. In particular, these mutations are restricted to the stromal component.[19][20]
-
Fibroadenoma histology (H&E). The image demonstrates intracanalicular morphology (bottom left) and pericanalicular morphology (top right)
-
Fibroadenoma, fine needle aspiration biopsy (Giemsa or DiffQuick stain). The image shows abundant bare bipolar stromal nuclei surrounding sheets of metachromatic epithelial cells.
-
Fibroadenoma, fine needle aspiration biopsy (Papanicolou stain). The image shows a sheet of epithelial cells in the typical antler pattern.
-
Histopathologic image of breast fibroadenoma. Core needle biopsy. Hematoxylin & eosin stain.
-
Histopathologic image of breast fibroadenoma showing proliferation of intralobular stroma compressing and distorting the epithelium. H&E stain.
-
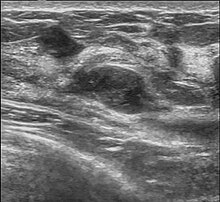
Fibroadenoma in ultrasound
-
Treatment
Fibroadenomas can be expected to shrink naturally, so most are simply monitored.[21] Monitoring fibroadenomas involves regular check-ups to make sure that the breast mass is not growing and is not potentially cancerous.[21] Check-ups involve physical examinations performed every 3–6 months and optional diagnostic imaging performed every 6–12 months for 1–2 years.[21] Generally, surgery is only recommended if the fibroadenoma gets larger or causes increased symptoms.[22] They are removed with a small margin of normal breast tissue if the preoperative clinical investigations are suggestive of the necessity of this procedure. A small amount of normal tissue must be removed in case the lesion turns out to be a phyllodes tumour on microscopic examination.[11][23]
Because needle biopsy is often a reliable diagnostic investigation, some doctors may decide not to operate to remove the lesion, and instead opt for clinical follow-up to observe the lesion over time using clinical examination and mammography to determine the rate of growth, if any, of the lesion. A growth rate of less than sixteen percent per month in women under fifty years of age, and a growth rate of less than thirteen percent per month in women over fifty years of age have been published as safe growth rates for continued non-operative treatment and clinical observation.[24]
Some fibroadenomas respond to treatment with ormeloxifene.[25]
Fibroadenomas have not been shown to recur following complete excision or transform into phyllodes tumours following partial or incomplete excision.[11]
Interventions
There are several non-invasive options for the treatment of fibroadenomas, including percutaneous radiofrequency ablation (RFA), cryoablation, and percutaneous microwave ablation.[26] With the use of advanced medical imaging, these procedures do not require invasive surgery and have the potential for enhanced cosmetic results compared with conventional surgery.[26]
Cryoablation
The FDA approved cryoablation of a fibroadenoma as a safe, effective, and minimally-invasive alternative to open surgical removal in 2001.[27] During cryoablation, ultrasound imaging is used to guide a probe into the mass of breast tissue. Extremely cold temperatures are then used to destroy the abnormal cells, and over time the cells are reabsorbed into the body.[28] The procedure can be performed as an outpatient surgery using local anesthesia, and leaves substantially less scarring than open surgical procedures and no breast tissue deformation.[citation needed]
The American Society of Breast Surgeons recommends the following criteria to establish a patient as a candidate for cryoablation of a fibroadenoma:[27]
- The lesion must be sonographically visible.
- The diagnosis of a fibroadenoma must be confirmed histologically.
- The lesion should be less than 4 cm in diameter.
High-intensity focused ultrasound
High-intensity focused ultrasound (HIFU) is a technique for the treatment of tumors of the breast and has shown promising results in the form of complete radiological removal of tumors.[29] An ultrasound beam is focused on a target in the breast and leads to tissue death and protein degradation by raising the temperature in that area.[29] Currently, the use of radiation is recommended in some cases, but HIFU in particular is not part of treatment guidelines.[30] Further research into the usefulness of HIFU, specifically in fibroadenoma, is required before more widespread use of the technique in fibroadenoma.[29]
Epidemiology
Of all breast tissue samples taken, fibroadenomas comprise about 50%, and this rate rises to 75% for tissue sample in women under the age of 20 years.[31] Fibroadenomas are more frequent among women in higher socioeconomic classes and darker-skinned people.[31] Body mass index and the number of full-term pregnancies were found to have a negative correlation with the risk of fibroadenomas.[31] There are no known genetic factors that influence the rate of fibroadenomas.[31] The rate of occurrence of fibroadenomas in women have been reported in literature to range from 7% to 13%.[31]
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 Salati, SA (7 December 2020). "Breast fibroadenomas: a review in the light of current literature". Polski przeglad chirurgiczny. 93 (1): 40–48. doi:10.5604/01.3001.0014.5676. PMID 33729177.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 "Breast Health: Follow-up after an abnormal mammogram - NCI". www.cancer.gov. 30 October 2014. Archived from the original on 5 September 2023. Retrieved 19 October 2023.
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 3.11 3.12 3.13 Stachs, A; Stubert, J; Reimer, T; Hartmann, S (9 August 2019). "Benign Breast Disease in Women". Deutsches Arzteblatt international. 116 (33–34): 565–574. doi:10.3238/arztebl.2019.0565. PMID 31554551.
- ↑ Oprić, S; Oprić, D; Gugić, D; Granić, M (March 2012). "Phyllodes tumors and fibroadenoma common beginning and different ending". Collegium antropologicum. 36 (1): 235–41. PMID 22816226.
- ↑ 5.0 5.1 5.2 5.3 5.4 5.5 5.6 "Fibroadenomas of the Breast". cancer.org. Archived from the original on 13 November 2018. Retrieved 13 November 2018.
- ↑ Shin SJ, Rosen PP (July 2007). "Bilateral presentation of fibroadenoma with digital fibroma-like inclusions in the male breast". Archives of Pathology & Laboratory Medicine. 131 (7): 1126–9. doi:10.5858/2007-131-1126-BPOFWD. PMID 17617003.
- ↑ Pruthi S, Jones KN (February 2013). "Nonsurgical management of fibroadenoma and virginal breast hypertrophy". Seminars in Plastic Surgery. 27 (1): 62–6. doi:10.1055/s-0033-1343997. PMC 3706058. PMID 24872742.
- ↑ Nelson ZC, Ray RM, Wu C, Stalsberg H, Porter P, Lampe JW, Shannon J, Horner N, Li W, Wang W, Hu Y, Gao D, Thomas DB (July 2010). "Fruit and vegetable intakes are associated with lower risk of breast fibroadenomas in Chinese women". The Journal of Nutrition. 140 (7): 1294–301. doi:10.3945/jn.109.119719. PMC 2884330. PMID 20484549.
- ↑ 9.0 9.1 DeMay, M. (2007). Practical Principles of Cytopathology (Revised ed.). ASCP Press. p. 2007. ISBN 978-0-89189-549-7.[page needed]
- ↑ Tasoulis, MK. "Evaluation of breast mass". BMJ Best Practice. Archived from the original on 13 November 2018. Retrieved 13 November 2018.
- ↑ 11.0 11.1 11.2 11.3 11.4 Rosen, PP. (2009). Rosen's Breast Pathology (3rd ed.). ISBN 978-0-7817-7137-5.[page needed]
- ↑ Fekete P, Petrek J, Majmudar B, Someren A, Sandberg W (May 1987). "Fibroadenomas with stromal cellularity. A clinicopathologic study of 21 patients". Archives of Pathology & Laboratory Medicine. 111 (5): 427–32. PMID 3032124.
- ↑ Nassar, Hind. "Cellular fibroadenoma of breast". Archived from the original on 21 October 2012. Retrieved 14 January 2013.
- ↑ Yang, Xiaofang; Kandil, Dina; Cosar, Ediz F.; Khan, Ashraf (2014). "Fibroepithelial Tumors of the Breast: Pathologic and Immunohistochemical Features and Molecular Mechanisms". Archives of Pathology & Laboratory Medicine. 138 (1): 25–36. doi:10.5858/arpa.2012-0443-RA. ISSN 0003-9985. PMID 24377809.
- ↑ "Fibroadenoma". Patholines.org. Archived from the original on 2022-05-23. Retrieved 2023-08-21. This page was last edited on 4 November 2019
- ↑ Tavassoli, F.A.; Devilee, P., eds. (2003). World Health Organization Classification of Tumours: Pathology & Genetics: Tumours of the breast and female genital organs. Lyon: IARC Press. ISBN 978-92-832-2412-9.[page needed]
- ↑ Pathology Outlines Website. [1] Archived 2018-09-07 at the Wayback Machine Accessed 12 February 2009.
- ↑ "Fibroadenoma of the breast". Archived from the original on 10 March 2009. Retrieved 15 December 2007.
- ↑ Lim WK, Ong CK, Tan J, Thike AA, Ng CC, Rajasegaran V, Myint SS, Nagarajan S, Nasir ND, McPherson JR, Cutcutache I, Poore G, Tay ST, Ooi WS, Tan VK, Hartman M, Ong KW, Tan BK, Rozen SG, Tan PH, Tan P, Teh BT (August 2014). "Exome sequencing identifies highly recurrent MED12 somatic mutations in breast fibroadenoma". Nature Genetics. 46 (8): 877–80. doi:10.1038/ng.3037. PMID 25038752. S2CID 7623650.
- ↑ Piscuoglio S, Murray M, Fusco N, Marchiò C, Loo FL, Martelotto LG, Schultheis AM, Akram M, Weigelt B, Brogi E, Reis-Filho JS (November 2015). "MED12 somatic mutations in fibroadenomas and phyllodes tumours of the breast". Histopathology. 67 (5): 719–29. doi:10.1111/his.12712. PMC 4996373. PMID 25855048.
- ↑ 21.0 21.1 21.2 American College of Obstetricians Gynecologists' Committee on Practice Bulletins—Gynecology (June 2016). "Practice Bulletin No. 164: Diagnosis and Management of Benign Breast Disorders". Obstetrics and Gynecology. 127 (6): e141-56. doi:10.1097/aog.0000000000001482. PMID 27214189. S2CID 37836506.
- ↑ "Fibroadenoma – Diagnosis and treatment – Mayo Clinic". mayoclinic.org. Archived from the original on 14 November 2018. Retrieved 13 November 2018.
- ↑ Rosai, J. (2004). Rosai and Ackerman's Surgical Pathology (9th ed.). ISBN 978-0-323-01342-0.[page needed]
- ↑ Gordon PB, Gagnon FA, Lanzkowsky L (October 2003). "Solid breast masses diagnosed as fibroadenoma at fine-needle aspiration biopsy: acceptable rates of growth at long-term follow-up". Radiology. 229 (1): 233–8. doi:10.1148/radiol.2291010282. PMID 14519878.
- ↑ Dhar A, Srivastava A (June 2007). "Role of centchroman in regression of mastalgia and fibroadenoma". World Journal of Surgery. 31 (6): 1178–84. doi:10.1007/s00268-007-9040-4. PMID 17431715. S2CID 22828570.
- ↑ 26.0 26.1 Sag AA, Maybody M, Comstock C, Solomon SB (June 2014). "Percutaneous image-guided ablation of breast tumors: an overview". Seminars in Interventional Radiology. 31 (2): 193–202. doi:10.1055/s-0034-1376159. PMC 4078155. PMID 25049447.
- ↑ 27.0 27.1 "Management of Fibroadenomas of the Breast : Official Statement" (PDF). American Society of Breast Surgeons. April 29, 2008. Archived (PDF) from the original on June 24, 2020. Retrieved June 23, 2020.
- ↑ "WebMD – Cryotherapy Shrinks Benign Breast Lumps". Archived from the original on 2014-02-08. Retrieved 2023-08-21.
- ↑ 29.0 29.1 29.2 Peek MC, Wu F (2018). "High-intensity focused ultrasound in the treatment of breast tumours". ecancermedicalscience. 12: 794. doi:10.3332/ecancer.2018.794. PMC 5804717. PMID 29434660.
- ↑ "BMJ Best Practice". bestpractice.bmj.com. Archived from the original on 13 November 2018. Retrieved 13 November 2018.
- ↑ 31.0 31.1 31.2 31.3 31.4 Greenberg R, Skornick Y, Kaplan O (September 1998). "Management of breast fibroadenomas". Journal of General Internal Medicine. 13 (9): 640–5. doi:10.1046/j.1525-1497.1998.cr188.x. PMC 1497021. PMID 9754521.
External links
| Classification | |
|---|---|
| External resources |