Faricimab
 | |
| Monoclonal antibody | |
|---|---|
| Type | Whole antibody |
| Source | Humanized |
| Target | VEGF-A, angiopoietin 2[1] |
| Names | |
| Trade names | Vabysmo |
| Other names | RO6867461; RG7716; faricimab-svoa |
| Clinical data | |
| Main uses | Wet age-related macular degeneration (nAMD), diabetic macular edema (DME)[1] |
| Side effects | Conjunctival bleeding, eye infection[1] |
| Routes of use | Intravitreal |
| Typical dose | 6 mg[1] |
| External links | |
| AHFS/Drugs.com | Monograph |
| Legal | |
| License data | |
| Legal status | |
| Chemical and physical data | |
| Formula | C6506H9968N1724O1026S45 |
| Molar mass | 130197.05 g·mol−1 |
Faricimab, sold under the brand name Vabysmo, is a medication used to treat wet age-related macular degeneration (nAMD) and diabetic macular edema (DME).[1] It is given by injection into the eye.[1]
Common side effects include conjunctival bleeding.[1] Other side effects may include eye infection and allergic reactions.[1] Use is not recommended in pregnancy.[1] It is a monoclonal antibody, that blocks both vascular endothelial growth factor (VEGF) and angiopoietin 2 (Ang-2), thus decreasing new blood vessel growth in the eye.[1][2]
Faricimab was approved for medical use in the United States and Europe in 2022.[1][2] In the United Kingdom a 28.8 mg vial costs the NHS about £860.[6] In the United States 6 mg costs about 2,300 USD as of 2022.[7]
Medical uses
Faricimab is indicated for treatment of neovascular age-related macular degeneration (nAMD) and diabetic macular edema (DME).[1]
Dosage
It is generally given at a dose of 6 mg every 4 weeks for 4 doses after which it may be given less frequently.[1]
Side effects
The most common side effect is conjunctival bleeding.[1]
Contraindications
Contraindications to injection of faricimab include active infection in or around the eye, active inflammation in the eye (uveitis), and prior allergic reactions to receiving the drug (hypersensitivity).[1]
Pregnancy and breastfeeding
There are no adequate and well-controlled studies of faricimab administration in pregnant women.[1] No studies on the effects of faricimab on human fertility have been conducted and it is not known whether faricimab can affect reproduction. Based on its mechanism of action, treatment with may pose a risk to reproductive capacity.[1]
There is no information regarding faricimab accumulation in breast milk, the effects of the drug on the breastfed infant, or the effects of the drug on milk production. However, the drug company states that many drugs are transferred in human milk with the potential for absorption and adverse reactions in the breastfed child.[1]
Pharmacology
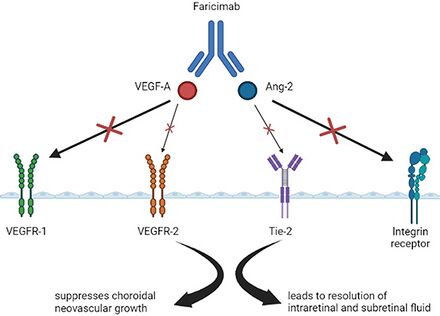
Faricimab is a 150kDa-sized bispecific antibody whose molecular structure allows a high affinity bond to both vascular endothelial growth factor A (VEGF-A) and Angiopoietin (Ang-2).[9]
By blocking the action of these two growth factors, faricimab decreases migration and replication of endothelial cells allowing for stabilization of vascular structures, thereby decreasing vascular leakage.[10][11][12] Faricimab has shown improved and sustained efficacy in comparison to agents that only target the VEGF pathway.[10]
History
In 2016, pre-clinical studies looking at the mechanism of action behind faricimab showed that by blocking Ang-2, one of the drug's targets, there was decreased endothelial barrier breakdown in blood vessels.[10] In 2017, phase I studies in neovascular age related macular degeneration (nAMD) showed that the drug was safe to use in people and well tolerated.[10]
Society and culture
Legal status
On 21 July 2022, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) adopted a positive opinion, recommending the granting of a marketing authorization for the medicinal product Vabysmo, intended for the treatment of neovascular age-related macular degeneration (nAMD) and diabetic macular oedema (DME).[13] The applicant for this medicinal product is Roche Registration GmbH.[13] Faricimab was approved for medical use in the European Union in September 2022.[2]
Names
Faricimab is the International Nonproprietary Name (INN).[14] Faricimab was formerly named RG7716.[15]
Research
Two phase II trials evaluated faricimab's efficacy and safety in comparison to ranibizumab and showed that faricimab received every 16 weeks and every twelve weeks was comparable to ranibizumab received every four weeks in visual acuity and imaging outcomes.[10] In 2019, two phase III multi-center randomized studies were initiated on 1200 participants with neovascular age related macular degeneration (nAMD) to evaluate faricimab's safety, efficacy, and durability against aflibercept.[10]
Diabetic macular edema
One phase II trial evaluated faricimab's efficacy and safety in comparison to ranibizumab and showed clinically meaningful and statistically significant improvements in visual acuity.[10][16] Two phase III multi-center randomized studies were completed on 1,891 diabetic participants with diabetic macular edema (DME)[10][17]
Retinal vein occlusion macular edema
In two phase III trials, faricimab met the primary endpoint of non-inferior visual acuity gains when compared with aflibercept in 553 participants with macular edema due to branch retinal vein occlusion (BRVO) and central retinal vein occlusion (CRVO) in the BALATON and COMINO studies.[18][19][20]
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 1.14 1.15 1.16 1.17 1.18 "Vabysmo- faricimab injection, solution". DailyMed. 7 February 2022. Archived from the original on 21 February 2022. Retrieved 20 February 2022.
- ↑ 2.0 2.1 2.2 2.3 "Vabysmo EPAR". European Medicines Agency (EMA). 19 July 2022. Archived from the original on 14 October 2022. Retrieved 14 October 2022.
- ↑ "Archive copy". Archived from the original on 2022-10-13. Retrieved 2022-10-28.
{{cite web}}: CS1 maint: archived copy as title (link) - ↑ "Summary Basis of Decision - Vabysmo". Health Canada. 31 August 2022. Archived from the original on 29 September 2022. Retrieved 29 September 2022.
- ↑ "Vabysmo Product information". Health Canada. 25 April 2012. Archived from the original on 1 October 2022. Retrieved 30 September 2022.
- ↑ "Faricimab". SPS - Specialist Pharmacy Service. 16 November 2018. Archived from the original on 11 December 2022. Retrieved 11 December 2022.
- ↑ "Vabysmo Prices, Coupons, Copay & Patient Assistance". Drugs.com. Retrieved 11 December 2022.
- ↑ Panos, Georgios D.; Lakshmanan, Arun; Dadoukis, Panagiotis; Ripa, Matteo; Motta, Lorenzo; Amoaku, Winfried M. (18 September 2023). "Faricimab: Transforming the Future of Macular Diseases Treatment - A Comprehensive Review of Clinical Studies". Drug Design, Development and Therapy. 17: 2861–2873. doi:10.2147/DDDT.S427416.
- ↑ Nicolò M, Ferro Desideri L, Vagge A, Traverso CE (March 2021). "Faricimab: an investigational agent targeting the Tie-2/angiopoietin pathway and VEGF-A for the treatment of retinal diseases". Expert Opinion on Investigational Drugs. 30 (3): 193–200. doi:10.1080/13543784.2021.1879791. PMID 33471572. S2CID 231665201.
- ↑ 10.0 10.1 10.2 10.3 10.4 10.5 10.6 10.7 Khan M, Aziz AA, Shafi NA, Abbas T, Khanani AM (August 2020). "Targeting Angiopoietin in Retinal Vascular Diseases: A Literature Review and Summary of Clinical Trials Involving Faricimab". Cells. 9 (8): 1869. doi:10.3390/cells9081869. PMC 7464130. PMID 32785136.
- ↑ Iglicki M, González DP, Loewenstein A, Zur D (February 2022). "Next-generation anti-VEGF agents for diabetic macular oedema". Eye. 36 (2): 273–277. doi:10.1038/s41433-021-01722-8. PMC 8807622. PMID 34373607. Archived from the original on 30 July 2022. Retrieved 30 July 2022.
{{cite journal}}: CS1 maint: PMC embargo expired (link) - ↑ Fenner BJ, Cheung CM, Sim SS, Lee WK, Staurenghi G, Lai TY, et al. (February 2022). "Evolving treatment paradigms for PCV". Eye. 36 (2): 257–265. doi:10.1038/s41433-021-01688-7. PMC 8807588. PMID 34262165. Archived from the original on 30 July 2022. Retrieved 30 July 2022.
- ↑ 13.0 13.1 "Vabysmo: Pending EC decision". European Medicines Agency. 22 July 2022. Archived from the original on 28 July 2022. Retrieved 29 July 2022. Text was copied from this source which is copyright European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ↑ World Health Organization (2018). "International nonproprietary names for pharmaceutical substances (INN): recommended INN: list 80". WHO Drug Information. 32 (3). hdl:10665/330907.
- ↑ Kaplon H, Chenoweth A, Crescioli S, Reichert JM (January 2022). "Antibodies to watch in 2022". mAbs. 14 (1): 2014296. doi:10.1080/19420862.2021.2014296. PMC 8765076. PMID 35030985.
- ↑ "Phase II data support potential for Roche's novel anti-VEGF/anti-angiopoietin-2 bispecific antibody, RG7716, for people with diabetic macular edema". Roche (Press release). Archived from the original on 17 February 2022. Retrieved 17 February 2022.
- ↑ Sharma A, Kumar N, Parachuri N, Karanam D, Kuppermann BD, Bandello F, Regillo CD (April 2022). "Faricimab phase 3 DME trial significance of personalized treatment intervals (PTI) regime for future DME trials". Eye. 36 (4): 679–680. doi:10.1038/s41433-021-01831-4. PMC 8956679. PMID 34718339. S2CID 240157515.
{{cite journal}}: CS1 maint: PMC embargo expired (link) - ↑ Hoffmann-La Roche (2 September 2022). "A Study to Evaluate the Efficacy and Safety of Faricimab in Participants With Macular Edema Secondary to Branch Retinal Vein Occlusion (BALATON)". Archived from the original on 28 October 2022. Retrieved 28 October 2022.
{{cite journal}}: Cite journal requires|journal=(help) - ↑ Hoffmann-La Roche (30 August 2022). "A Study to Evaluate the Efficacy and Safety of Faricimab in Participants With Macular Edema Secondary to Central Retinal or Hemiretinal Vein Occlusion (COMINO)". Archived from the original on 28 October 2022. Retrieved 28 October 2022.
{{cite journal}}: Cite journal requires|journal=(help) - ↑ "Roche touts positive top-line Phase III results for Vabysmo". The Pharma Letter. Archived from the original on 28 October 2022. Retrieved 28 October 2022.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- Clinical trial number NCT03622580 for "A Study to Evaluate the Efficacy and Safety of Faricimab (RO6867461) in Participants With Diabetic Macular Edema (YOSEMITE)" at ClinicalTrials.gov
- Clinical trial number NCT03622593 for "A Study to Evaluate the Efficacy and Safety of Faricimab (RO6867461) in Participants With Diabetic Macular Edema (RHINE)" at ClinicalTrials.gov
- Clinical trial number NCT03823287 for "A Study to Evaluate the Efficacy and Safety of Faricimab in Participants With Neovascular Age-Related Macular Degeneration (TENAYA)" at ClinicalTrials.gov
- Clinical trial number NCT03823300 for "A Study to Evaluate the Efficacy and Safety of Faricimab in Participants With Neovascular Age-Related Macular Degeneration (LUCERNE)" at ClinicalTrials.gov
- Pages using duplicate arguments in template calls
- CS1 maint: archived copy as title
- CS1 maint: PMC embargo expired
- CS1 errors: missing periodical
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Drugs missing an ATC code
- Articles containing unverified chemical infoboxes
- Drugs that are a monoclonal antibody
- Drugs not assigned an ATC code
- Angiogenesis inhibitors
- Genentech brands
- Hoffmann-La Roche brands
- Monoclonal antibodies
- Ophthalmology drugs
- RTT