Ethambutol
 | |
 Chemical structure of ethambutol (top) and photo of ethambutol crystals (bottom) | |
| Names | |
|---|---|
| Trade names | Myambutol, Etibi,[1] Servambutol, others |
| Other names | (2S,2’S)-2,2’-(Ethane-1,2-diyldiimino)dibutan-1-ol[2] |
| Clinical data | |
| Main uses | Tuberculosis[3] |
| Pregnancy category |
|
| Routes of use | By mouth |
| Defined daily dose | 1,200 mg (by mouth) 1,200 mg (parenteral)[5] |
| External links | |
| AHFS/Drugs.com | Monograph |
| Legal | |
| License data |
|
| Legal status |
|
| Pharmacokinetics | |
| Protein binding | 20–30% |
| Metabolism | liver |
| Elimination half-life | 3–4 hours |
| Chemical and physical data | |
| Formula | C10H24N2O2 |
| Molar mass | 204.314 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Ethambutol (EMB, E) is a medication primarily used to treat tuberculosis.[4] It is usually given in combination with other tuberculosis medications, such as isoniazid, rifampicin and pyrazinamide.[6] It may also be used to treat Mycobacterium avium complex, and Mycobacterium kansasii.[4] It is taken by mouth.[4]
Common side effects include problems with vision, joint pain, nausea, headaches, and feeling tired.[4] Other side effects include liver problems and allergic reactions.[4] It is not recommended in people with optic neuritis, significant kidney problems, or under the age of five.[6] Use during pregnancy or breastfeeding has not been found to cause harm.[6][7] In the United States the FDA has raised concerns about eye issues in the baby if used during pregnancy.[4] Ethambutol is believed to work by interfering with the bacteria's metabolism.[4]
Ethambutol was discovered in 1961.[8] It is on the World Health Organization's List of Essential Medicines.[9] Ethambutol is available as a generic medication.[1] The wholesale cost in the developing world is about US$2.58–4.73 per month.[10] In the United States it costs US$100–200 per month.[1]
Medical uses
Ethambutol is used along with other medications to treat a number of infections including: tuberculosis, Mycobacterium avium complex, and Mycobacterium kansasii.[4]
Dosage
The defined daily dose is 1,200 mg (by mouth) or 1,200 mg (parenteral).[5] The dose is 15 to 25 mg/kg once per day to a maximum of 1,200 mg.[3]
Side effects
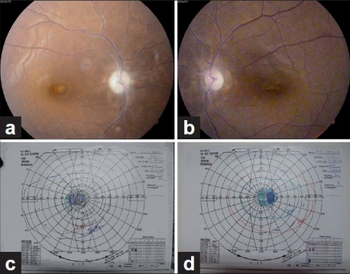
- Optic neuritis[11] (hence contraindicated in children below six years of age)
- Red-green color blindness[12][13] People taking ethambutol should be monitored for changes in visual acuity and color discrimination.[12]
- Arthralgia
- Hyperuricaemia
- Vertical nystagmus
- Milk skin reaction
Mechanism of action
Ethambutol is bacteriostatic against actively growing TB bacilli. It works by obstructing the formation of cell wall. Mycolic acids attach to the 5'-hydroxyl groups of D-arabinose residues of arabinogalactan and form mycolyl-arabinogalactan-peptidoglycan complex in the cell wall. It disrupts arabinogalactan synthesis by inhibiting the enzyme arabinosyl transferase. Disruption of the arabinogalactan synthesis inhibits the formation of this complex and leads to increased permeability of the cell wall.
Pharmacokinetics
It is well absorbed from the gastrointestinal tract and well distributed in body tissues and fluids. 50% is excreted unchanged in urine.
References
- ↑ 1.0 1.1 1.2 Hamilton, Richart (2015). Tarascon Pocket Pharmacopoeia 2015 Deluxe Lab-Coat Edition. Jones & Bartlett Learning. p. 48. ISBN 9781284057560.
- ↑ "ethambutol (CHEBI:4877)". Chemical Entities of Biological Interest. UK: European Bioinformatics Institute. 18 August 2010. Main. Archived from the original on 19 July 2014. Retrieved 26 April 2012.
- ↑ 3.0 3.1 "ETHAMBUTOL = E oral - Essential drugs". medicalguidelines.msf.org. Archived from the original on 28 August 2021. Retrieved 30 August 2020.
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 4.7 4.8 "Ethambutol Hydrochloride". The American Society of Health-System Pharmacists. Archived from the original on 5 June 2016. Retrieved 10 December 2016.
- ↑ 5.0 5.1 "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 27 November 2020. Retrieved 21 September 2020.
- ↑ 6.0 6.1 6.2 World Health Organization (2009). Stuart MC, Kouimtzi M, Hill SR (eds.). WHO Model Formulary 2008. World Health Organization. pp. 136, 138, 588, 603. hdl:10665/44053. ISBN 9789241547659.
- ↑ "Prescribing medicines in pregnancy database". Australian Government. 3 March 2014. Archived from the original on 8 April 2014. Retrieved 22 April 2014.
- ↑ Landau, Ralph; Achilladelis, Basil; Scriabine, Alexander (1999). Pharmaceutical Innovation: Revolutionizing Human Health. Chemical Heritage Foundation. p. 171. ISBN 9780941901215. Archived from the original on 2016-12-20.
- ↑ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ↑ "Ethambutol". International Drug Price Indicator Guide. Archived from the original on 22 January 2018. Retrieved 8 December 2016.
- ↑ Lim SA (April 2006). "Ethambutol-associated optic neuropathy" (PDF). Ann. Acad. Med. Singap. 35 (4): 274–8. PMID 16710500. Archived (PDF) from the original on 2009-08-16.
- ↑ 12.0 12.1 Lewis, Sharon Mantik; Dirksen, Shannon Ruff; Heitkemper, Margaret M.; Bucher, Linda; Harding, Mariann. Medical-surgical nursing : assessment and management of clinical problems (9th ed.). St. Louis, Missouri. ISBN 978-0-323-10089-2. OCLC 228373703.
- ↑ Tripathi, K D (August 2015). Essentials of MEDICAL PHARMACOLOGY (Seventh ed.). India: JAYPEE BROTHERS MEDICAL PUBLISHERS. p. 769. ISBN 978-93-5025-937-5.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- "Ethambutol". Medicine Plus. Archived from the original on 2008-10-01. Retrieved 2016-05-10.
- Pages using duplicate arguments in template calls
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Articles containing unverified chemical infoboxes
- Anti-tuberculosis drugs
- Diamines
- Diols
- World Health Organization essential medicines
- RTT