Epidemic

An epidemic (from Greek ἐπί epi "upon or above" and δῆμος demos "people") is the rapid spread of disease to a large number of hosts in a given population within a short period of time. For example, in meningococcal infections, an attack rate in excess of 15 cases per 100,000 people for two consecutive weeks is considered an epidemic.[1][2]
Epidemics of infectious disease are generally caused by several factors including a change in the ecology of the host population (e.g., increased stress or increase in the density of a vector species), a genetic change in the pathogen reservoir or the introduction of an emerging pathogen to a host population (by movement of pathogen or host). Generally, an epidemic occurs when host immunity to either an established pathogen or newly emerging novel pathogen is suddenly reduced below that found in the endemic equilibrium and the transmission threshold is exceeded.[3]
An epidemic may be restricted to one location; however, if it spreads to other countries or continents and affects a substantial number of people, it may be termed a pandemic.[1]: §1:72 The declaration of an epidemic usually requires a good understanding of a baseline rate of incidence; epidemics for certain diseases, such as influenza, are defined as reaching some defined increase in incidence above this baseline.[2] A few cases of a very rare disease may be classified as an epidemic, while many cases of a common disease (such as the common cold) would not. An epidemic can cause enormous damage through financial and economic losses in addition to impaired health and loss of life.[citation needed]
Definition
The United States Centers for Disease Control and Prevention defines epidemic broadly: "Epidemic refers to an increase, often sudden, in the number of cases of a disease above what is normally expected in that population in that area." The term "outbreak" can also apply, but is usually restricted to smaller events.[1]: §1:72 [2]
Any sudden increase in disease prevalence may generally be termed an epidemic. This may include contagious disease (i.e. easily spread between persons) such as influenza; vector-borne diseases such as malaria; water-borne diseases such as cholera; and sexually transmitted diseases such as HIV/AIDS. The term can also be used for non-communicable health issues such as obesity.[2][4][5]
The term epidemic derives from a word form attributed to Homer's Odyssey, which later took its medical meaning from the Epidemics, a treatise by Hippocrates.[5] Before Hippocrates, epidemios, epidemeo, epidamos, and other variants had meanings similar to the current definitions of "indigenous" or "endemic".[5] Thucydides' description of the Plague of Athens is considered one of the earliest accounts of a disease epidemic.[5] By the early 17th century, the terms endemic and epidemic referred to contrasting conditions of population-level disease, with the endemic condition a "common sicknesse" and the epidemic "hapning in some region, or countrey, at a certaine time, ....... producing in all sorts of people, one and the same kind of sicknesse".[6]
The term "epidemic" is often applied to diseases in non-human animals, although "epizootic" is technically preferable.[7][8]
-
Map of West Africa showing the number of days since last confirmed case of Ebola virus disease in region and the number of confirmed cases in past 21 days during January 25-February 14, 2015[9]
-
Western African Ebola virus epidemic synopsis
-
Example of an epidemic showing the number of new infections over time.
Causes
There are several factors that may contribute (individually or in combination) to causing an epidemic. There may be changes in a pathogen, in the population that it can infect, in the environment, or in the interaction between all three. Factors include:[10]: §1:72
Antigenic Change
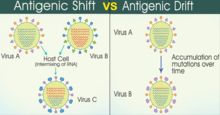
An antigen is a protein on the virus' surface that host antibodies can recognize and attack. Changes in the antigenic characteristics of the agent make it easier for the changed virus to spread throughout a previously immune population. There are two natural mechanisms for change - antigenic drift and antigenic shift. Antigenic drift arises over a period of time as an accumulation of mutations in the virus genes, possibly through a series of hosts, and eventually gives rise to a new strain of virus which can evade existing immunity. Antigenic shift is abrupt - in this, two or more different strains of a virus, coinfecting a single host, combine to form a new subtype having a mixture of characteristics of the original strains. The best known and best documented example of both processes is influenza.[11] SARS-CoV2 has demonstrated antigenic drift and possibly shift as well.[12]
Drug resistance
Antibiotic resistance applies specifically to bacteria that become resistant to antibiotics.[13] Resistance in bacteria can arise naturally by genetic mutation, or by one species acquiring resistance from another through horizontal gene transfer.[14] Extended use of antibiotics appears to encourage selection for mutations which can render antibiotics ineffective. This is especially true of tuberculosis, with increasing occurrence of multiple drug-resistant tuberculosis (MDR-TB) worldwide.[15][16]
Changes in transmission

Pathogen transmission is a term used to describe the mechanisms by which a disease-causing agent (virus, bacterium, or parasite) spreads from one host to another. Common modes of transmission include:[17] -
- Airborne (as with influenza and COVID-19),
- Fecal-oral (as with cholera and typhoid),
- Vector-borne (malaria, Zika) and
- Sexual (syphilis, HIV)
The first three of these require that pathogen must survive away from its host for a period of time; an evolutionary change which increases survival time will result in increased virulence.[18]
Another possibility, although rare, is that a pathogen may adapt to take advantage of a new mode of transmission[19][20]
Seasonality
Seasonal diseases arise due to the change in the environmental conditions, especially such as humidity and temperature, during different seasons. Many diseases display seasonality,[21][22] This may be due to one or more of the following underlying factors: -[23]
- the ability of the pathogen to survive outside the host - e.g. water-borne cholera[24] which becomes prevalent in tropical wet seasons, or influenza which peaks in temperate regions during winter.[25][26]
- The behaviour of people susceptible to the disease - such as spending more time in close contact indoors.[27]
- Changes in immune function during winter - one possibility is a reduction in vitamin D, and another is the effect of cold on mucus membranes in the nose.[28][29]
- Abundance of vectors such as mosquitoes.[30]
Human behaviour

Changes in behaviour can affect the likelihood or severity of epidemics. The classic example is the 1854 Broad Street cholera outbreak, in which a cholera outbreak was mitigated by removing a supply of contaminated water - an event now regarded as the foundation of the science of epidemiology.[31] Urbanisation and overcrowding (e.g. in refugee camps) increase the likelihood of disease outbreaks.[32][33] A factor which contributed to the initial rapid increase in the 2014 Ebola virus epidemic was ritual bathing of (infective) corpses; one of the control measures was an education campaign to change behaviour around funeral rites.[34]
Changes in host population
The level of immunity to a disease in a population - herd immunity - is at its peak after a disease outbreak or a vaccination campaign. In the following years, immunity will decline, both within individuals and in the population as a whole as older individuals die and new individuals are born. Eventually, unless there is another vaccination campaign, an outbreak or epidemic will recur.[35]
It's also possible for disease which is endemic in one population to become epidemic if it is introduced into a novel setting where the host population is not immune. An example of this was the introduction European diseases such as smallpox into indigenous populations during the 16th century.[36]
Zoonosis
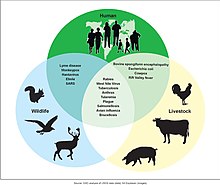
A zoonosis is an infectious disease of humans caused by a pathogen that can jump from a non-human host to a human.[37]
Major diseases such as Ebola virus disease and salmonellosis are zoonoses. HIV was a zoonotic disease transmitted to humans in the early part of the 20th century, though it has now evolved into a separate human-only disease.[38]
Some strains of bird flu and swine flu are zoonoses; these viruses occasionally recombine with human strains of the flu and can cause pandemics such as the 1918 Spanish flu or the 2009 swine flu.[39]
Types
Common source outbreak
In a common source outbreak epidemic, the affected individuals had an exposure to a common agent. If the exposure is singular and all of the affected individuals develop the disease over a single exposure and incubation course, it can be termed a point source outbreak. If the exposure was continuous or variable, it can be termed a continuous outbreak or intermittent outbreak, respectively.[1]: 56
Propagated outbreak
In a propagated outbreak, the disease spreads person-to-person. Affected individuals may become independent reservoirs leading to further exposures.[1]: 56 Many epidemics will have characteristics of both common source and propagated outbreaks (sometimes referred to as mixed outbreak).[citation needed]
For example, secondary person-to-person spread may occur after a common source exposure or an environmental vector may spread a zoonotic diseases agent.[1]: 56–58
Preparation
Preparations for an epidemic include having a disease surveillance system; the ability to quickly dispatch emergency workers, especially local-based emergency workers; and a legitimate way to guarantee the safety and health of health workers.[40][41]
Effective preparations for a response to a pandemic are multi-layered. The first layer is a disease surveillance system. Tanzania, for example, runs a national lab that runs testing for 200 health sites and tracks the spread of infectious diseases. The next layer is the actual response to an emergency. According to U.S.-based columnist Michael Gerson in 2015, only the U.S. military and NATO have the global capability to respond to such an emergency.[40] Still, despite the most extensive preparatory measures, a fast-spreading pandemic may easily exceed and overwhelm existing health-care resources.[42] Consequently, early and aggressive mitigation efforts, aimed at the so-called "epidemic curve flattening" need to be taken.[42] Such measures usually consist on non-pharmacological interventions such as social/physical distancing, aggressive contact tracing, "stay-at-home" orders, as well as appropriate personal protective equipment (i.e., masks, gloves, and other physical barriers to spread).[42]
See also
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 Dicker, Richard C.; Coronado, Fátima; Koo, Denise; Parrish, R. Gibson (2012). Principles of epidemiology in public health practice; an introduction to applied epidemiology and biostatistics. 3rd ed (Third ed.). Atlanta, Georgia: Centers for Disease Control and Prevention. Archived from the original on 2023-10-01. Retrieved 2024-01-25.
- ↑ 2.0 2.1 2.2 2.3 Green MS, Swartz T, Mayshar E, Lev B, Leventhal A, Slater PE, Shemer J (January 2002). "When is an epidemic an epidemic?" (PDF). The Israel Medical Association Journal. 4 (1): 3–6. PMID 11802306. Archived (PDF) from the original on 2020-11-28. Retrieved 2024-01-25.
- ↑ Callow PP, ed. (1998). "Epidemic". The Encyclopedia of Ecology and Environmental Management. Oxford: Blackwell Science Ltd. p. 246. ISBN 0-86542-838-7. Archived from the original on 2024-02-12. Retrieved 2024-01-25.
- ↑ Controlling the global obesity epidemic Archived 2020-10-27 at the Wayback Machine, the World Health Organization
- ↑ 5.0 5.1 5.2 5.3 Martin PM, Martin-Granel E (June 2006). "2,500-year evolution of the term epidemic". Emerging Infectious Diseases. 12 (6): 976–80. doi:10.3201/eid1206.051263. PMC 3373038. PMID 16707055.
- ↑ Lodge, Thomas (1603). A treatise of the plague: containing the nature, signes, and accidents of the same, with the certaine and absolute cure of the fevers, botches and carbuncles that raigne in these times. London: Edward White.
CHAP. 1. Of the nature and essence of the Plague
- ↑ McKie, Robin (2021-02-21). "Foot and mouth 20 years on: what an animal virus epidemic taught UK science". The Observer. ISSN 0029-7712. Archived from the original on 2023-09-17. Retrieved 2023-09-11.
- ↑ "Emergency response for epizootic diseases". Agri-Food and Biosciences Institute. 11 December 2005. Archived from the original on 27 September 2023. Retrieved 11 September 2023.
- ↑ "Update: Ebola Virus Disease Epidemic — West Africa, February 2015". www.cdc.gov. Archived from the original on 31 January 2024. Retrieved 31 January 2024.
- ↑ Dicker, Richard C.; Coronado, Fátima; Koo, Denise; Parrish, R. Gibson (2012). Principles of epidemiology in public health practice; an introduction to applied epidemiology and biostatistics. 3rd ed (Third ed.). Atlanta, Georgia: Centers for Disease Control and Prevention. Archived from the original on 2023-10-01. Retrieved 2024-01-25.
- ↑ CDC (12 December 2022). "How Flu Viruses Can Change". Centers for Disease Control and Prevention. Archived from the original on 18 March 2021. Retrieved 9 September 2023.
- ↑ Carabelli, Alessandro M.; Peacock, Thomas P.; Thorne, Lucy G.; Harvey, William T.; Hughes, Joseph; de Silva, Thushan I.; Peacock, Sharon J.; Barclay, Wendy S.; de Silva, Thushan I.; Towers, Greg J.; Robertson, David L. (March 2023). "SARS-CoV-2 variant biology: immune escape, transmission and fitness". Nature Reviews Microbiology. 21 (3): 162–177. doi:10.1038/s41579-022-00841-7. ISSN 1740-1534. PMID 36653446.
- ↑ "Antimicrobial resistance Fact sheet N°194". who.int. April 2014. Archived from the original on 10 March 2015. Retrieved 7 March 2015.
- ↑ "General Background: About Antibiotic Resistance". www.tufts.edu. Archived from the original on 23 October 2015. Retrieved 30 October 2015.
- ↑ "Tuberculosis (TB)". who.int. Archived from the original on 30 July 2020. Retrieved 8 May 2020.
- ↑ Dabour R, Meirson T, Samson AO (December 2016). "Global antibiotic resistance is mostly periodic". Journal of Global Antimicrobial Resistance. 7: 132–134. doi:10.1016/j.jgar.2016.09.003. PMID 27788414.
- ↑ "FAQ: Methods of Disease Transmission". Department of Microbiology, Mount Sinai Hospital. Archived from the original on 17 March 2020. Retrieved 10 January 2024.
- ↑ Mandavilli, Apoorva (1 October 2021). "Is the Coronavirus Getting Better at Airborne Transmission?". The New York Times. ISSN 0362-4331. Archived from the original on 24 September 2023. Retrieved 12 September 2023.
- ↑ Alcamí, Antonio (2023-03-28). "Pathogenesis of the circulating mpox virus and its adaptation to humans". Proceedings of the National Academy of Sciences. 120 (13): e2301662120. Bibcode:2023PNAS..12001662A. doi:10.1073/pnas.2301662120. ISSN 0027-8424. PMID 36940331.
- ↑ Antonovics, Janis; Wilson, Anthony J.; Forbes, Mark R.; Hauffe, Heidi C.; Kallio, Eva R.; Leggett, Helen C.; Longdon, Ben; Okamura, Beth; Sait, Steven M.; Webster, Joanne P. (2017-05-05). "The evolution of transmission mode". Philosophical Transactions of the Royal Society B: Biological Sciences. 372 (1719): 20160083. doi:10.1098/rstb.2016.0083. ISSN 0962-8436. PMC 5352810. PMID 28289251.
- ↑ Martinez, Micaela Elvira (8 November 2018). "The calendar of epidemics: Seasonal cycles of infectious diseases". PLOS Pathogens. 14 (11): e1007327. doi:10.1371/journal.ppat.1007327. ISSN 1553-7374. PMC 6224126. PMID 30408114.
- ↑ "Mark Your Calendar: All Infectious Diseases Are Seasonal". Columbia University Mailman School of Public Health. 8 November 2018. Archived from the original on 2 October 2023. Retrieved 13 September 2023.
- ↑ Grassly, Nicholas C; Fraser, Christophe (7 October 2006). "Seasonal infectious disease epidemiology". Proceedings of the Royal Society B: Biological Sciences. 273 (1600): 2541–2550. doi:10.1098/rspb.2006.3604. ISSN 0962-8452. PMC 1634916. PMID 16959647.
- ↑ Leitzell, Katherine (20 November 2011). "The Time of Cholera". NASA Earthdata. Archived from the original on 11 November 2023. Retrieved 25 January 2024.
- ↑ CDC (2022-09-20). "Learn more about the flu season". Centers for Disease Control and Prevention. Retrieved 2023-09-13.
{{cite web}}: CS1 maint: url-status (link) - ↑ Marr, Linsey C.; Tang, Julian W.; Van Mullekom, Jennifer; Lakdawala, Seema S. (January 2019). "Mechanistic insights into the effect of humidity on airborne influenza virus survival, transmission and incidence". Journal of the Royal Society Interface. 16 (150): 20180298. doi:10.1098/rsif.2018.0298. ISSN 1742-5689. PMC 6364647. PMID 30958176.
- ↑ Robson, David (19 October 2015). "The real reason germs spread in the winter". www.bbc.com. Archived from the original on 22 September 2023. Retrieved 14 September 2023.
- ↑ Kashef, Ziba (2019-05-13). "Flu virus' best friend: low humidity". YaleNews. Archived from the original on 2023-10-02. Retrieved 2023-09-13.
- ↑ LaMotte, Sandee (6 December 2022). "Scientists finally know why people get more colds and flu in winter". CNN. Archived from the original on 2023-10-02. Retrieved 2023-09-14.
- ↑ Medicine, Institute of; Health, Board on Global; Threats, Forum on Microbial (2008-03-18). Vector-Borne Diseases: Understanding the Environmental, Human Health, and Ecological Connections: Workshop Summary. National Academies Press. ISBN 978-0-309-17770-2. Archived from the original on 2023-10-03. Retrieved 2024-01-25.
- ↑ Tulchinsky, Theodore H. (2018). "John Snow, Cholera, the Broad Street Pump; Waterborne Diseases Then and Now". Case Studies in Public Health: 77–99. doi:10.1016/B978-0-12-804571-8.00017-2. ISBN 9780128045718. PMC 7150208.
- ↑ Neiderud, Carl-Johan (2015-06-24). "How urbanization affects the epidemiology of emerging infectious diseases". Infection Ecology & Epidemiology. 5 (1): 10.3402/iee.v5.27060. Bibcode:2015InfEE...527060N. doi:10.3402/iee.v5.27060. ISSN 2000-8686. PMC 4481042. PMID 26112265.
- ↑ Altare, Chiara; Kahi, Vincent; Ngwa, Moise; Goldsmith, Amelia; Hering, Heiko; Burton, Ann; Spiegel, Paul (1 September 2019). "Infectious disease epidemics in refugee camps: a retrospective analysis of UNHCR data (2009-2017)". Journal of Global Health Reports. 3: e2019064. doi:10.29392/joghr.3.e2019064. S2CID 207998081. Archived from the original on 11 November 2023. Retrieved 25 January 2024.
- ↑ Maxmen, Amy (30 January 2015). "How the Fight Against Ebola Tested a Culture's Traditions". National Geographic. Archived from the original on March 8, 2021. Retrieved 14 September 2023.
- ↑ Yang, Luojun; Grenfell, Bryan T; Mina, Michael J (February 2020). "Waning immunity and re-emergence of measles and mumps in the vaccine era". Current Opinion in Virology. 40: 48–54. doi:10.1016/j.coviro.2020.05.009. PMID 32634672. S2CID 220414525. Archived from the original on 2022-06-20. Retrieved 2024-01-25.
- ↑ "Stacy Goodling, "Effects of European Diseases on the Inhabitants of the New World"". Archived from the original on 10 May 2008.
- ↑ "Zoonoses". World Health Organization. 29 July 2020. Archived from the original on 23 March 2022. Retrieved 14 September 2023.
- ↑ Sharp PM, Hahn BH (September 2011). "Origins of HIV and the AIDS pandemic". Cold Spring Harbor Perspectives in Medicine. 1 (1): a006841. doi:10.1101/cshperspect.a006841. PMC 3234451. PMID 22229120.
- ↑ Scotch M, Brownstein JS, Vegso S, Galusha D, Rabinowitz P (September 2011). "Human vs. animal outbreaks of the 2009 swine-origin H1N1 influenza A epidemic". EcoHealth. 8 (3): 376–380. doi:10.1007/s10393-011-0706-x. PMC 3246131. PMID 21912985.
- ↑ 40.0 40.1 Gerson M (26 March 2015). "The next epidemic". The Washington Post. Archived from the original on 26 March 2023. Retrieved 25 January 2024.
- ↑ Gates B (April 2015). "The next epidemic--lessons from Ebola". The New England Journal of Medicine. 372 (15): 1381–4. doi:10.1056/NEJMp1502918. PMID 25853741.
- ↑ 42.0 42.1 42.2 Stawicki SP, Jeanmonod R, Miller AC, Paladino L, Gaieski DF, Yaffee AQ, et al. (2020). "The 2019-2020 Novel Coronavirus (Severe Acute Respiratory Syndrome Coronavirus 2) Pandemic: A Joint American College of Academic International Medicine-World Academic Council of Emergency Medicine Multidisciplinary COVID-19 Working Group Consensus Paper". Journal of Global Infectious Diseases. 12 (2): 47–93. doi:10.4103/jgid.jgid_86_20. PMC 7384689. PMID 32773996. S2CID 218754925.
Further reading
- Brook, Timothy; et al. "Comparative pandemics: the Tudor–Stuart and Wanli–Chongzhen years of pestilence, 1567–1666" Journal of Global History (2020) 14#3 pp 363–379 emphasis on Chinese history, compared to England
- Eisenberg, Merle, and Lee Mordechai. "The Justinianic Plague and Global Pandemics: The Making of the Plague Concept." American Historical Review 125.5 (2020): 1632–1667.
- Honigsbaum, Mark (18 October 2020). "How do pandemics end? In different ways, but it's never quick and never neat". The Guardian. ISSN 0261-3077. Archived from the original on 27 October 2020. Retrieved 28 October 2020.
- Lietaert Peerbolte, Bert Jan (September 2021). "The Book of Revelation: Plagues as Part of the Eschatological Human Condition". Journal for the Study of the New Testament. SAGE Publications. 44 (1): 75–92. doi:10.1177/0142064X211025496. ISSN 1745-5294. S2CID 237332665.
- McKenna, Maryn, "Return of the Germs: For more than a century drugs and vaccines made astounding progress against infectious diseases. Now our best defenses may be social changes", Scientific American, vol. 323, no. 3 (September 2020), pp. 50–56. "What might prevent or lessen [the] possibility [of a virus emerging and finding a favorable human host] is more prosperity more equally distributed – enough that villagers in South Asia need not trap and sell bats to supplement their incomes and that, low-wage workers in the U.S. need not go to work while ill because they have no sick leave." (p. 56.)
- "Escaping the 'Era of Pandemics': Experts Warn Worse Crises to Come Options Offered to Reduce Risk". Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services. 2020. Archived from the original on 2024-01-26. Retrieved 2024-01-25.
External links
| Look up epidemic in Wiktionary, the free dictionary. |
- "European Centre for Disease Prevention and Control". Archived from the original on 2020-06-02. Retrieved 2024-01-25.
- "International Epidemiological Association (IEA)". Archived from the original on 2010-11-27.
- A Dictionary of Epidemiology (IEA). Oxford University Press. 20 June 2014. ISBN 978-0-19-997672-0. Archived from the original on 2 October 2023. Retrieved 25 January 2024.
- "People's Epidemiology Library". Archived from the original on 2012-03-23.
- "Video Discussion of the Prostate Cancer Epidemic". Archived from the original on 2016-03-03. Retrieved 2024-01-25.
- "Simulations of epidemic spread across a landscape". Monash Virtual Laboratory. Archived from the original on 2007-11-04.
![Map of West Africa showing the number of days since last confirmed case of Ebola virus disease in region and the number of confirmed cases in past 21 days during January 25-February 14, 2015[9]](https://upload.wikimedia.org/wikipedia/commons/thumb/c/c4/M6407a6f.gif/449px-M6407a6f.gif)
