Enterotoxigenic Escherichia coli
| Enterotoxigenic Escherichia coli | |
|---|---|
| Other names: ETEC, Enterotoxigenic E coli infection[1] | |
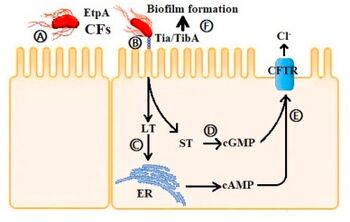 | |
| a-f)Adherence mechanism of ETEC[2] | |
| Specialty | Infectious disease |
Enterotoxigenic Escherichia coli (ETEC) is a type of Escherichia coli and one of the leading bacterial causes of diarrhea in the developing world,[3] as well as the most common cause of travelers' diarrhea.[4] Insufficient data exists, but conservative estimates suggest that each year, about 157,000 deaths occur, mostly in children, from ETEC.[5][6][7] A number of pathogenic isolates are termed ETEC, but the main hallmarks of this type of bacteria are expression of one or more enterotoxins and presence of fimbriae used for attachment to host intestinal cells. The bacteria was identified by the Bradley Sack lab in Kolkata in 1968.
Enterotoxin types
Enterotoxins produced by ETEC include heat-labile enterotoxin (LT) and heat-stable enterotoxin (ST).[8]
Signs and symptoms
Infection with ETEC can cause profuse, watery diarrhea with no blood or leukocytes and abdominal cramping. Fever, nausea with or without vomiting, chills, loss of appetite, headache, muscle aches and bloating can also occur, but are less common.[4]
Transmission
This infection is transmitted via contaminated food or water.[9]
Diagnosis
In terms of the diagnosis we find that bacteria are cultured from samples of stool, as well as based on a individual’s history and symptoms, are used in the evaluation process[9]
Prevention
To date, no licensed vaccines specifically target ETEC, though several are in various stages of development.[3][11] Studies indicate that protective immunity to ETEC develops after natural or experimental infection, suggesting that vaccine-induced ETEC immunity should be feasible and could be an effective preventive strategy.[3][12] Prevention through vaccination is a critical part of the strategy to reduce the incidence and severity of diarrheal disease due to ETEC, particularly among children in low-resource settings. The development of a vaccine against this infection has been hampered by technical constraints, insufficient support for coordination, and a lack of market forces for research and development. Most vaccine development efforts are taking place in the public sector or as research programs within biotechnology companies.[13] ETEC is a longstanding priority and target for vaccine development for the World Health Organization.[citation needed]
Management
Treatment for ETEC infection includes rehydration therapy and antibiotics, although ETEC is frequently resistant to common antibiotics.[4] Improved sanitation is also key. Since the transmission of this bacterium is fecal contamination of food and water supplies, one way to prevent infection is by improving public and private health facilities. Another simple prevention of infection is by drinking factory bottled water—this is especially important for travelers and traveling military—though it may not be feasible in developing countries, which carry the greatest disease burden.[citation needed]
See also
References
- ↑ "Enterotoxigenic E coli Infection (Concept Id: C4763883) - MedGen - NCBI". www.ncbi.nlm.nih.gov. Archived from the original on 22 December 2022. Retrieved 29 May 2023.
- ↑ Govindarajan, Deenadayalan Karaiyagowder; Viswalingam, Nandhini; Meganathan, Yogesan; Kandaswamy, Kumaravel (1 September 2020). "Adherence patterns of Escherichia coli in the intestine and its role in pathogenesis". Medicine in Microecology. 5: 100025. doi:10.1016/j.medmic.2020.100025. ISSN 2590-0978. Archived from the original on 3 April 2023. Retrieved 21 February 2024.
- ↑ 3.0 3.1 3.2 Bourgeois, A Louis; Wierzba, Thomas F; Walker, Richard I (2016). "Status of vaccine research and development for enterotoxigenic Escherichia coli". Vaccine. 34 (26): 2880–2886. doi:10.1016/j.vaccine.2016.02.076. PMID 26988259.
- ↑ 4.0 4.1 4.2 US Centers for Disease Control and Prevention (2014). "Enterotoxigenic E. coli (ETEC)". Archived from the original on 2023-03-06. Retrieved 2023-02-04.
- ↑ Lozano, Rafael; Naghavi, Mohsen; Foreman, Kyle; et al. (2012). "Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010". The Lancet. 380 (9859): 2095–2128. doi:10.1016/S0140-6736(12)61728-0. hdl:10536/DRO/DU:30050819. PMID 23245604. S2CID 1541253. Archived from the original on 2022-11-23. Retrieved 2023-02-04.
- ↑ Gupta, SK; Keck J; Ram PK; et al. (2008). "Analysis of Data Gaps Pertaining to Enterotoxigenic Escherichia coli Infections in Low and Medium Human Development Index Countries, 1984-2005". Epidemiology and Infection. 136 (6): 721–738. doi:10.1017/S095026880700934X. PMC 2870873. PMID 17686197.
- ↑ Kotloff, Karen L; Nataro, James P; Blackwelder, William C; et al. (2013). "Burden and aetiology of diarrhoeal disease in infants and young children in developing countries (the Global Enteric Multicenter Study, GEMS): a prospective, case-control study". The Lancet. 382 (9888): 209–222. doi:10.1016/s0140-6736(13)60844-2. PMID 23680352. S2CID 205969172. Archived from the original on 2021-04-23. Retrieved 2023-02-04.
- ↑ Qadri, F.; Svennerholm, A. M.; Faruque, A. S. G.; Bradley Sack, R. (2005). "Enterotoxigenic Escherichia coli in Developing Countries: Epidemiology, Microbiology, Clinical Features, Treatment, and Prevention". Clinical Microbiology Reviews. 18 (3): 465–483. doi:10.1128/CMR.18.3.465-483.2005. PMC 1195967. PMID 16020685.
- ↑ 9.0 9.1 "Enterotoxigenic E. coli (ETEC) | E. coli | CDC". www.cdc.gov. 14 January 2019. Archived from the original on 12 May 2023. Retrieved 29 May 2023.
- ↑ Caeiro, Juan‐Pablo; Estrada‐Garcia, M. Teresa; Jiang, Zhi‐Dong; Mathewson, John J.; Adachi, Javier A.; Steffen, Robert; DuPont, Herbert L. (December 1999). "Improved Detection of Enterotoxigenic Escherichia coli among Patients with Travelers' Diarrhea, by Use of the Polymerase Chain Reaction Technique". The Journal of Infectious Diseases. 180 (6): 2053–2055. doi:10.1086/315121. Retrieved 21 February 2024.
- ↑ "WHO vaccine pipeline tracker". World Health Organization. Archived from the original on March 22, 2016. Retrieved 2016-07-21.
- ↑ Girard, M.; Steele, D.; Chaignat, C. L.; Kieny, M. P. (2006). "A review of vaccine research and development: human enteric infections". Vaccine. 24 (15): 2732–2750. doi:10.1016/j.vaccine.2005.10.014. PMID 16483695.
- ↑ PATH, bvgh (March 2011). "The Case for Investment in Enterotoxigenic Escherichia coli Vaccines" (PDF). Archived (PDF) from the original on 29 July 2013. Retrieved 2 May 2012.
{{cite journal}}: Cite journal requires|journal=(help)
External links
| Classification |
|---|