Doxapram
 | |
| Names | |
|---|---|
| Trade names | Dopram, Stimulex, Respiram, others |
| Other names | Doxapram hydrochloride |
| |
| Clinical data | |
| Drug class | Respiratory stimulant[1] |
| Main uses | Respiratory depression, acute respiratory failure[1] |
| Side effects | Cough, shortness of breath, headache, dizziness, high blood pressure, flushing, sweating, vomiting, muscle spasms[2] |
| Routes of use | Intravenous |
| Onset of action | 30 sec[2] |
| Duration of action | 5 to 12 min[2] |
| External links | |
| AHFS/Drugs.com | Monograph |
| Legal | |
| Legal status |
|
| Chemical and physical data | |
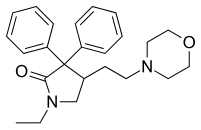
| Formula | C24H30N2O2 |
| Molar mass | 378.516 g·mol−1 |
Doxapram, sold under the brand name Dopram among others, is a medication used to treat respiratory depression after surgery or acute respiratory failure.[1] It is not; however, a preferred treatment.[2] It is given by injection into a vein.[1] Effects generally occur around 30 seconds and last for around 10 minutes.[2]
Common side effects include cough, shortness of breath, headache, dizziness, high blood pressure, flushing, sweating, vomiting, and muscle spasms.[2] Other side effects may include QT prolongation, seizure, and red blood cell breakdown.[2] While there is no evidence of harm in pregnancy, the manufacturer does not recommend.[1] It is a respiratory stimulant.[1]
Doxapram was approved for medical use in the United States in 1965.[2] It is available as a generic medication.[1] In the United Kingdom 5 vials of 100 mg costs the NHS about £130 as of 2021.[1] This amount in the United States is about 72 USD.[3]
Medical uses
Doxapram is used in intensive care settings to stimulate the respiratory rate in patients with respiratory failure. It may be useful for treating respiratory depression in patients who have taken excessive doses of drugs such as buprenorphine or fentanyl analogues which may fail to respond adequately to treatment with naloxone.[4]
It is equally as effective as pethidine in suppressing shivering after surgery.[5]
Side effects
Side effects include high blood pressure, panic attacks, rapid heart rate, tremor, sweating, and vomiting. Convulsions have been reported. Its use is relatively contraindicated in people with coronary heart disease, epilepsy, and high blood pressure. It is also contraindicated in newborns and small children, mainly due to the presence of benzyl alcohol, which is included as a preservative.
Mechanism of action

Doxapram stimulates chemoreceptors in the carotid bodies of the carotid arteries, which in turn, stimulates the respiratory centre in the brain stem.
Chemistry
Doxapram is a white to off-white, odorless, crystalline powder that is stable in light and air. It is soluble in water, sparingly soluble in alcohol and practically insoluble in ether. Injectable products have a pH from 3.5-5. Benzyl alcohol or chlorobutanol is added as a preservative agent in the commercially available injections.
See also
- Pentethylcyclanone (similar structure)
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 315. ISBN 978-0857114105.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 "Doxapram Monograph for Professionals". Drugs.com. Archived from the original on 23 January 2021. Retrieved 27 December 2021.
- ↑ "Dopram Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 27 January 2021. Retrieved 27 December 2021.
- ↑ "Buprenorphine Drug Data Sheet" (PDF). Archived (PDF) from the original on 2018-09-20. Retrieved 2020-12-06.
- ↑ Singh P, Dimitriou V, Mahajan RP, Crossley AW (November 1993). "Double-blind comparison between doxapram and pethidine in the treatment of postanaesthetic shivering". British Journal of Anaesthesia. 71 (5): 685–8. doi:10.1093/bja/71.5.685. PMID 8251281.
External links
| Identifiers: |
|
|---|
- Pages using duplicate arguments in template calls
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Drugs missing an ATC code
- Respiratory agents
- Pyrrolidones
- Morpholines
- RTT