Defibrotide
| Names | |
|---|---|
| Trade names | Defitelio |
| Other names | Defibrotide sodium |
| Clinical data | |
| Drug class | Oligonucleotides[1] |
| Main uses | Veno-occlusive disease of the liver[2] |
| Side effects | Low blood pressure, nausea, bleeding[3][4] |
| Pregnancy category | |
| Routes of use | Intravenous |
| Typical dose | 6.25 mg/kg QID[1] |
| External links | |
| AHFS/Drugs.com | Monograph |
| Legal | |
| License data | |
| Legal status | |
| Pharmacokinetics | |
| Bioavailability | 58 - 70% by mouth (i.v. and i.m. = 100%) |
| Elimination half-life | < 2 hours[4] |
Defibrotide, sold under the brand name Defitelio, is a medication used to treat veno-occlusive disease of the liver following bone marrow transplant.[2] It is given by injection into a vein.[2] High quality evidence to support its use is lacking.[2]
Common side effects include low blood pressure, nausea, and bleeding.[3][4] Other side effects may include allergic reactions.[4] It should not be used in people on blood thinners.[4] It is a mixture of oligonucleotides and is believed to work in part by decreasing endothelial cell activation.[1]
Defibrotide was approved for medical use in Europe in 2013, the United States in 2016, and Australia in 2020.[3][2][5] In the United States 25 doses of 200 mg cost about 10,000 USD.[7]
Medical uses
Defibrotide is used to treat veno-occlusive disease of the liver of people having had a bone marrow transplant, with different limitations in the US and the European Union.[4][1] As of 2016, however, randomized placebo controlled trials have not been done.[8]
In the European Union defibrotide is indicated for the treatment of severe hepatic veno-occlusive disease (VOD) also known as sinusoidal obstructive syndrome (SOS) in hematopoietic stem-cell transplantation (HSCT) therapy for people over one month of age.[3]
Hematopoietic stem cell transplantation (HSCT) is a procedure performed in some people to treat certain blood or bone marrow cancers.[9] Immediately before an HSCT procedure, a patient receives chemotherapy.[9] Hepatic VOD can occur in people who receive chemotherapy and HSCT.[9] Hepatic VOD is a condition in which some of the veins in the liver become blocked, causing swelling and a decrease in blood flow inside the liver, which may lead to liver damage.[9] In the most severe form of hepatic VOD, the patient may also develop failure of the kidneys and lungs.[9] Fewer than two percent of people develop severe hepatic VOD after HSCT, but as many as 80 percent of people who develop severe hepatic VOD do not survive.[9]
Dosage
A dose of 6.25 mg/kg four times per day may also be used.[1] This is given for a least 3 weeks.[1]
It is administered by intravenous infusion in a doctor's office or clinic.[1][3]
Contraindications
Use of defibrotide for people who are already taking anticoagulants is dangerous and use of other drugs that affect platelet aggregation, like NSAIDs, should be done with care. Defibrotide should not be given to people who have a difficult time maintaining a steady blood pressure.[1]
Side effects
There is a high risk of bleeding and some people have had hypersensitivity reactions to defibrotide.[4][1]
Common adverse effects, occurring in between 1 and 10% of people, included impaired blood clotting, vomiting, low blood pressure, bleeding in the brain, eyes, lungs, stomach or intestines, in the urine, and at catheterization sites.[1]
Other side effects have included diarrhea, nosebleeds, sepsis, graft vs host disease, and pneumonia.[4]
Pregnant women should not take defibrotide and women should not become pregnant while taking it; it has not been tested in pregnant women but at normal doses it caused hemolytic abortion in rats.[1]
Pharmacology
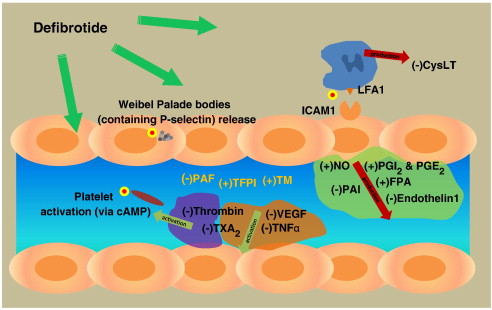
Defibrotide's mechanism of action is poorly understood. In vitro studies have shown that it protects the endothelium lining blood vessels from damage by fludarabine, a chemotherapy drug, and from a few other insults like serum starvation. It also appears to increase t-PA function and decrease plasminogen activator inhibitor-1 activity.[4][1]
Chemistry
Defibrotide is a mixture of single-stranded oligonucleotides. The chemical name is polydeoxyribonucleotide, sodium salt.[4] It is purified from the intestinal mucosa of pigs.[1]
History
The efficacy of defibrotide was investigated in 528 participants treated in three studies: two prospective clinical trials and an expanded access study.[9] The participants enrolled in all three studies had a diagnosis of hepatic VOD with liver or kidney abnormalities after hematopoietic stem cell transplantation (HSCT).[9] The studies measured the percentage of participants who were still alive 100 days after HSCT (overall survival).[9] In the three studies, 38 to 45 percent of participants treated with defibrotide were alive 100 days after HSCT.[9] Based on published reports and analyses of participant-level data, the expected survival rates 100 days after HSCT would be 21 to 31 percent for participants with severe hepatic VOD who received only supportive care or interventions other than defibrotide.[9] Defibrotide is the first FDA-approved therapy for treatment of severe hepatic VOD, a rare and life-threatening liver condition.[9]
Society and culture
Legal status
Defibrotide was approved in the European Union for use in treating veno-occlusive disease of the liver of people having had a bone marrow transplant in 2013;[3][11] Gentium had developed it.[12] At the end of that year, Jazz Pharmaceuticals acquired Gentium.[12]
In March 2016, the U.S. Food and Drug Administration (FDA) approved it for a similar use.[9][13][11] Defibrotide is the first FDA-approved therapy for treatment of severe hepatic VOD, a rare and life-threatening liver condition.[9] The FDA granted the application for defibrotide priority review status and orphan drug designation.[9] The FDA granted approval of Defitelio to Jazz Pharmaceuticals.[9]
Defibrotide was approved for medical use in Japan in June 2019.[14]
Defibrotide was approved for medical use in Australia in July 2020.[5]
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 "Defitelio 80 mg/mL concentrate for solution for infusion - Summary of Product Characteristics". UK Electronic Medicines Compendium. 26 May 2016. Archived from the original on 31 March 2018. Retrieved 20 July 2017.
- ↑ 2.0 2.1 2.2 2.3 2.4 "Defibrotide Monograph for Professionals". Drugs.com. Archived from the original on 21 December 2021. Retrieved 22 December 2021.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 "Defitelio EPAR". European Medicines Agency (EMA). Archived from the original on 28 October 2020. Retrieved 16 August 2020.
- ↑ 4.00 4.01 4.02 4.03 4.04 4.05 4.06 4.07 4.08 4.09 4.10 "Defitelio- defibrotide sodium injection, solution". DailyMed. 30 March 2016. Archived from the original on 29 October 2020. Retrieved 16 August 2020.
- ↑ 5.0 5.1 5.2 5.3 "Defitelio Australian Prescription Medicine Decision Summary". Therapeutic Goods Administration (TGA). 31 July 2020. Archived from the original on 13 August 2020. Retrieved 16 August 2020.
- ↑ "Defibrotide (Defitelio) Use During Pregnancy". Drugs.com. 6 July 2020. Archived from the original on 26 November 2020. Retrieved 16 August 2020.
- ↑ "Defitelio Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 15 April 2021. Retrieved 22 December 2021.
- ↑ Dalle JH, Giralt SA (March 2016). "Hepatic Veno-Occlusive Disease after Hematopoietic Stem Cell Transplantation: Risk Factors and Stratification, Prophylaxis, and Treatment". Biology of Blood and Marrow Transplantation. 22 (3): 400–9. doi:10.1016/j.bbmt.2015.09.024. PMID 26431626.
- ↑ 9.00 9.01 9.02 9.03 9.04 9.05 9.06 9.07 9.08 9.09 9.10 9.11 9.12 9.13 9.14 9.15 "FDA approves first treatment for rare disease in patients who receive stem cell transplant from blood or bone marrow". U.S. Food and Drug Administration (FDA) (Press release). March 30, 2016. Archived from the original on August 16, 2020. Retrieved August 21, 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ Pescador, R.; Capuzzi, L.; Mantovani, M.; Fulgenzi, A.; Ferrero, M. E. (1 July 2013). "Defibrotide: Properties and clinical use of an old/new drug". Vascular Pharmacology. 59 (1): 1–10. doi:10.1016/j.vph.2013.05.001. ISSN 1537-1891.
- ↑ 11.0 11.1 "Jazz Pharmaceuticals and Nippon Shinyaku Enter Into License Agreements for the Development and Commercialization of Defitelio and Vyxeos in Japan" (Press release). Jazz Pharmaceuticals plc. 30 March 2017. Archived from the original on 17 August 2020. Retrieved 16 August 2020 – via PR Newswire.
- ↑ 12.0 12.1 "Jazz Pharma Acquiring Gentium for $1B". GEN Genetic Engineering & Biotechnology News. December 20, 2013. Archived from the original on December 22, 2013. Retrieved August 21, 2020.
- ↑ "Defitelio Injection". U.S. Food and Drug Administration (FDA). 10 May 2016. Archived from the original on 8 April 2021. Retrieved 16 August 2020.
- Lay summary in: (PDF) https://www.accessdata.fda.gov/drugsatfda_docs/nda/2016/208114Orig1s000SumR.pdf.
{{cite web}}: Missing or empty|title=(help)
- Lay summary in: (PDF) https://www.accessdata.fda.gov/drugsatfda_docs/nda/2016/208114Orig1s000SumR.pdf.
- ↑ "Marketing Approval of Defitelio by MHLW for the Treatment of Sinusoidal Obstruction Syndrome / Hepatic Veno-Occlusive Disease" (Press release). Nippon Shinyaku Co., Ltd. 18 June 2019. Archived from the original on 13 August 2020. Retrieved 16 August 2020 – via CMOCRO.
External links
- Richardson P, Aggarwal S, Topaloglu O, Villa KF, Corbacioglu S (December 2019). "Systematic review of defibrotide studies in the treatment of veno-occlusive disease/sinusoidal obstruction syndrome (VOD/SOS)". Bone Marrow Transplant. 54 (12): 1951–1962. doi:10.1038/s41409-019-0474-8. PMC 6957462. PMID 30804485.
- Richardson PG, Carreras E, Iacobelli M, Nejadnik B (June 2018). "The use of defibrotide in blood and marrow transplantation". Blood Adv. 2 (12): 1495–1509. doi:10.1182/bloodadvances.2017008375. PMC 6020812. PMID 29945939.
| External sites: | |
|---|---|
| Identifiers: |
|
- Pages using duplicate arguments in template calls
- Wikipedia articles incorporating the PD-notice template
- CS1 errors: missing title
- CS1 errors: bare URL
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Infobox drug articles without a structure image
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Drugs missing an ATC code
- Drug has EMA link
- Drugboxes which contain changes to verified fields
- Chemicals that do not have a ChemSpider ID assigned
- Anticoagulants
- Orphan drugs
- RTT