Deferasirox
 | |
 | |
| Names | |
|---|---|
| Pronunciation | de FER a sir ox |
| Trade names | Exjade, Jadenu, others |
| Other names | CGP-72670, ICL-670A, IC L670 |
| |
| Clinical data | |
| Drug class | Iron chelator[1] |
| Main uses | Chronic iron overload due to blood transfusions[2] |
| Side effects | Kidney problems, nausea, diarrhea, heart burn, rash, itchiness, liver problems[2] |
| Pregnancy category |
|
| Routes of use | By mouth |
| Typical dose | 7 to 21 mg/kg OD[3] |
| External links | |
| AHFS/Drugs.com | Monograph |
| Legal | |
| License data |
|
| Legal status | |
| Pharmacokinetics | |
| Bioavailability | 70% |
| Protein binding | 99% |
| Metabolism | Liver glucuronidation |
| Elimination half-life | 8 to 16 hours |
| Excretion | Fecal (84%) and renal (8%) |
| Chemical and physical data | |
| Formula | C21H15N3O4 |
| Molar mass | 373.368 g·mol−1 |
| 3D model (JSmol) | |
| Density | 1.4±0.1 g/cm3 [4] |
| |
| |
Deferasirox, sold under the brand name Exjade among others, is a medication used for chronic iron overload in those receiving long-term blood transfusions for conditions such as beta-thalassemia and other chronic anemias.[2] It is generally only when deferoxamine is not sufficient.[2] It is taken by mouth.[3]
Common side effects include kidney problems, nausea, diarrhea, heart burn, rash, itchiness, and liver problems.[2] Other side effects may include metabolic acidosis, bone marrow suppression, and gastrointestinal bleeding.[2][1] It should not be used in people with kidney problems.[1] It is believed to be harmful in pregnancy.[5] It is an iron chelator.[1]
Deferasirox was approved for medical use in the United States in 2005 and Europe in 2006.[6][2] It is on the World Health Organization's List of Essential Medicines.[7] In the United Kingdom a month at a dose of 360 mg per day costs the NHS about £500 as of 2021.[3] In the United States this amount costs about 190 USD.[8] Generic versions were approved in 2020.[9]
Medical uses
It is approved in the European Union by the European Medicines Agency (EMA) for children six years and older for chronic iron overload from repeated blood transfusions.[10][11][12]
It may be used in those 2 years and over if deferoxamine is not sufficient.[2]
Dosage
It is typically started at a dose of 7 to 21 mg/kg per day.[3]
Side effects
Deferasirox ranked second on the list of drugs most frequently suspected in reported patient deaths compiled for 2019 by the Institute for Safe Medical Practices, with 1320 suspected deaths.[13] A boxed warning was added in the same year with regard to kidney failure, liver failure and gastrointestinal bleeding.[14] It is suspected that the main driver of this spike in suspected deaths relates to the re-analysis of adverse event data by Novartis.[13]
Properties
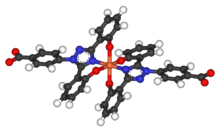
The half-life of deferasirox is between 8 and 16 hours allowing once a day dosing. Two molecules of deferasirox are capable of binding to 1 atom of iron which are subsequently eliminated by fecal excretion. Its low molecular weight and high lipophilicity allows the drug to be taken orally unlike deferoxamine which has to be administered by IV route (intravenous infusion). Together with deferiprone, deferasirox seems to be capable of removing iron from cells (cardiac myocytes and hepatocytes) as well as removing iron from the blood.
Synthesis
Deferasirox can be prepared from simple commercially available starting materials (salicylic acid, salicylamide and 4-hydrazinobenzoic acid) in the following two-step synthetic sequence:

The condensation of salicyloyl chloride (formed in situ from salicylic acid and thionyl chloride) with salicylamide under dehydrating reaction conditions results in formation of 2-(2-hydroxyphenyl)-1,3(4H)-benzoxazin-4-one. This intermediate is isolated and reacted with 4-hydrazinobenzoic acid in the presence of base to give 4-(3,5-bis(2-hydroxyphenyl)-1,2,4-triazol-1-yl)benzoic acid (deferasirox).[15]
Society and culture
In July 2020, Teva discontinued deferasirox.[16]
References
- ↑ 1.0 1.1 1.2 1.3 "DailyMed - DEFERASIROX- deferasirox tablet, film coated". dailymed.nlm.nih.gov. Archived from the original on 11 January 2022. Retrieved 21 December 2021.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 "Exjade". Archived from the original on 16 September 2021. Retrieved 21 December 2021.
- ↑ 3.0 3.1 3.2 3.3 BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 1074. ISBN 978-0857114105.
- ↑ "Material Safety Data Sheet (MSDS): Deferasirox". ChemSrc. 2018. Archived from the original on 2017-06-12. Retrieved 2021-06-28.
- ↑ "Deferasirox Use During Pregnancy". Drugs.com. Archived from the original on 29 November 2020. Retrieved 21 December 2021.
- ↑ "Deferasirox Monograph for Professionals". Drugs.com. Archived from the original on 15 August 2020. Retrieved 21 December 2021.
- ↑ World Health Organization (2023). The selection and use of essential medicines 2023: web annex A: World Health Organization model list of essential medicines: 23rd list (2023). Geneva: World Health Organization. hdl:10665/371090. WHO/MHP/HPS/EML/2023.02.
- ↑ "Deferasirox Prices and Deferasirox Coupons - GoodRx". GoodRx. Retrieved 21 December 2021.
- ↑ "Drugs@FDA: FDA-Approved Drugs". U.S. Food and Drug Administration. Archived from the original on 19 October 2020. Retrieved 15 August 2020.
- ↑ "Exjade – deferasirox" (PDF). European Medicines Agency. 2018. Archived (PDF) from the original on 2017-12-29. Retrieved 2021-06-28.
- ↑ Kontoghiorghes GJ (April 2013). "Turning a blind eye to deferasirox's toxicity?". Lancet. 381 (9873): 1183–4. doi:10.1016/S0140-6736(13)60799-0. PMID 23561999. S2CID 27794849.
- ↑ "Review: Exjade side effects". Archived from the original on 2016-03-04. Retrieved 2021-06-28.
- ↑ 13.0 13.1 "QuarterWatch™ (Quarter 4 and 2009 totals): Reported Patient Deaths Increased by 14% in 2009". Institute For Safe Medication Practices. Archived from the original on 2021-06-28. Retrieved 2021-06-28.
- ↑ Pediatrics, American Academy of (2010-02-19). "Black box warning added to Exjade". AAP News. doi:10.1542/aapnews.20100219-1. ISSN 1073-0397. Archived from the original on 2021-06-28. Retrieved 2021-06-28.
- ↑ Steinhauser S, Heinz U, Bartholomä M, Weyhermüller T, Nick H, Hegetschweiler K (2004). "Complex Formation of ICL670 and Related Ligands with FeIII and FeII". European Journal of Inorganic Chemistry. 2004 (21): 4177–4192. doi:10.1002/ejic.200400363.]
- ↑ "Deferasirox Discontinuation". U.S. Food and Drug Administration (FDA). Archived from the original on 21 July 2020. Retrieved 20 July 2020.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- Pages using duplicate arguments in template calls
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drug has EMA link
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- Chelating agents
- Chelating agents used as drugs
- Orphan drugs
- Novartis brands
- Antidotes
- Triazoles
- Benzoic acids
- Phenols
- RTT
- World Health Organization essential medicines