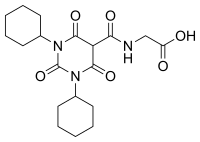
Daprodustat
 | |
| Names | |
|---|---|
| Trade names | Duvroq, Jesduvroq |
| Other names | GSK1278863 |
| |
| Clinical data | |
| Drug class | Hypoxia-inducible factor prolyl hydroxylase inhibitor[1] |
| Main uses | Low red blood cells due to chronic kidney disease[1] |
| Side effects | High blood pressure, blood clots, abdominal pain[1] |
| Routes of use | By mouth[1] |
| Typical dose | 1 to 24 mg OD[1] |
| External links | |
| AHFS/Drugs.com | Monograph |
| Legal | |
| License data |
|
| Legal status | |
| Chemical and physical data | |
| Formula | C19H27N3O6 |
| Molar mass | 393.440 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Daprodustat, sold under the brand name Duvroq among others, is a medication used to treat low red blood cells due to chronic kidney disease.[1] It is used in those who have been on dialysis for more than four months.[1] It is taken by mouth.[1] It is used to reduce the need for blood transfusions.[1]
Common side effects include high blood pressure, blood clots, and abdominal pain.[1] Other side effects may include worsening heart failure and upper gastrointestinal bleeding.[1] Use may increase the risk of death.[1] It is not recommended in those with active cancer.[1] It interacts with medications that affect CYP2C8.[1] Use in pregnancy may harm the baby.[1] It is a hypoxia-inducible factor prolyl hydroxylase inhibitor which works by increasing erythropoietin.[1][2]
Daprodustat was approved for medical use in Japan in 2020,[2] and the United States in 2023.[1] It is the first by mouth treatment for low red blood cells caused by kidney disease.[3][4] Previously injectable erythropoiesis-stimulating agents were used for this purpose.[5]
Medical uses
Daprodustat is indicated for the treatment of anemia due to chronic kidney disease.[1]
Dosage
It is taken once per day at a dose of 1 mg to 24 mg.[1]
History
Daprodustat increases erythropoietin levels.[3] The effectiveness of daprodustat was established in a randomized study of 2,964 adult participants receiving dialysis.[3] In this study, participants received either oral daprodustat or injected recombinant human erythropoietin (a standard of care treatment for people with anemia due to chronic kidney disease).[3] Daprodustat raised and maintained the hemoglobin (the protein in red blood cells that carries oxygen and is a common measure of anemia) within the target range of 10-11 grams/deciliter, similar to that of the recombinant human erythropoietin.[3] The US Food and Drug Administration (FDA) granted the approval of Jesduvroq to GlaxoSmithKline LLC.[3]
Society and culture
Due to its potential applications in athletic doping, it has also been incorporated into screens for performance-enhancing drugs.[6]
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 1.14 1.15 1.16 1.17 1.18 1.19 "Jesduvroq- daprodustat tablet, film coated". DailyMed. 1 February 2023. Archived from the original on 11 February 2023. Retrieved 11 February 2023.
- ↑ 2.0 2.1 Dhillon S (September 2020). "Daprodustat: First Approval". Drugs. 80 (14): 1491–1497. doi:10.1007/s40265-020-01384-y. PMC 7471535. PMID 32880805.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 "FDA Approves First Oral Treatment for Anemia Caused by Chronic Kidney Disease for Adults on Dialysis". U.S. Food and Drug Administration (FDA) (Press release). 1 February 2023. Archived from the original on 4 February 2023. Retrieved 3 February 2023.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ Harris, Emily (15 February 2023). "FDA Approves First Oral Treatment for Kidney Disease–Induced Anemia". Medical News in Brief. JAMA. 329 (9): 704. doi:10.1001/jama.2023.1556. PMID 36790833. Archived from the original on 19 March 2023. Retrieved 3 May 2023.
- ↑ "Issue 4 - March 1, 2023". www.pbdrx.com. Archived from the original on 30 June 2023. Retrieved 14 May 2023.
- ↑ Thevis M, Milosovich S, Licea-Perez H, Knecht D, Cavalier T, Schänzer W (August 2016). "Mass spectrometric characterization of a prolyl hydroxylase inhibitor GSK1278863, its bishydroxylated metabolite, and its implementation into routine doping controls". Drug Testing and Analysis. 8 (8): 858–63. doi:10.1002/dta.1870. PMID 26361079.
External links
| Identifiers: |
|
|---|
- Clinical trial number NCT02879305 for "Anemia Studies in Chronic Kidney Disease: Erythropoiesis Via a Novel Prolyl Hydroxylase Inhibitor Daprodustat-Dialysis (ASCEND-D)" at ClinicalTrials.gov
- Pages using duplicate arguments in template calls
- Wikipedia articles incorporating the PD-notice template
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Articles containing unverified chemical infoboxes
- Acetic acids
- Cyclohexyl compounds
- GSK plc brands
- Pyrimidinediones
- RTT
- All stub articles
- Blood and blood forming organ drug stubs