Copper deficiency
| Copper deficiency | |
|---|---|
| Other names: Hypocupremia | |
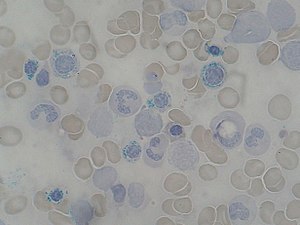 | |
| Ring Sideroblast smear, a sign of copper deficiency in the blood. | |
| Risk factors | Alcoholism, gastric bypass surgery |
Copper deficiency is defined either as insufficient copper to meet the needs of the body, or as a serum copper level below the normal range.[1] Copper deficiency can have blood related consequences, such as myelodysplasia, anemia, low white blood cell count, and low count of neutrophils.[2] It is also associated with neurological manifestations like sensory ataxia (irregular coordination due to proprioceptive loss), spasticity, muscle weakness, and more rarely visual loss due to damage in the peripheral nerves, myelopathy (disease of the spinal cord), and rarely optic neuropathy.
The most common cause is a remote gastrointestinal surgery, such as gastric bypass surgery, due to malabsorption of copper, or zinc toxicity. On the other hand, Menkes disease is a genetic disorder of copper deficiency involving a wide variety of symptoms that is often fatal.[3] Copper is required for the functioning of many enzymes, such as cytochrome c oxidase, which is complex IV in the mitochondrial electron transport chain, ceruloplasmin, Cu/Zn superoxide dismutase, and in amine oxidases.[4] These enzyme catalyze reactions for oxidative phosphorylation, iron transportation, antioxidant and free radical scavenging and neutralization, and neurotransmitter synthesis, respectively.[4] Diets vary in the amount of copper they contain, but may provide about 5 mg/day, of which only 20-50% is absorbed.[2] The diet of the elderly may have a lower copper content than the recommended daily intake.[2] Dietary copper can be found in whole grain cereals, legumes, oysters, organ meats (particularly liver), cherries, dark chocolate, fruits, leafy green vegetables, nuts, poultry, prunes, and soybean products like tofu.[5]
The deficiency has been recognized for some time in ruminant animals, in which it is commonly known as "swayback".[4] Copper deficiency can manifest in parallel with vitamin B12 and other nutritional deficiencies.[2]
Signs and symptoms
Blood
The characteristic hematological (blood) effects of copper deficiency are anemia (which may be microcytic, normocytic or macrocytic) and neutropenia.[6] Thrombocytopenia (low blood platelets) is unusual.[2][7]
The peripheral blood and bone marrow aspirate findings in copper deficiency can mimic myelodysplastic syndrome.[8] Bone marrow aspirate in both conditions may show dysplasia of blood cell precursors and the presence of ring sideroblasts (erythroblasts containing multiple iron granules around the nucleus). Unlike most cases of myelodysplastic syndrome, the bone marrow aspirate in copper deficiency characteristically shows cytoplasmic vacuoles within red and white cell precursors, and karyotyping in cases of copper deficiency does not reveal cytogenetic features characteristic of myelodysplastic syndrome.[6][7]
Anemia and neutropenia typically resolve within six weeks of copper replacement.[8]
Neurological
Copper deficiency can cause a wide variety of neurological problems including myelopathy, peripheral neuropathy, and optic neuropathy.[4][7]
Myelopathy
Copper deficiency myelopathy in humans was discovered and first described by Schleper and Stuerenburg in 2001.[9] They described a patient with a history of gastrectomy and partial colonic resection who presented with severe tetraparesis and painful paraesthesias and who was found on imaging to have dorsomedial cervical cord T2 hyperintensity. Upon further analysis, it was found that the patient had decreased levels of serum coeruloplasmin, serum copper, and CSF copper. The patient was treated with parenteral copper and the patient's paraesthesias did resolve. Since this discovery, there has been heightened and increasing awareness of copper-deficiency myelopathy and its treatment, and this disorder has been reviewed by Kumar. Patients typically present difficulty walking (gait difficulty) caused by sensory ataxia (irregular muscle coordination) due to dorsal column dysfunction[7] or degeneration of the spinal cord (myelopathy).[4][10] Patients with ataxic gait have problems balancing and display an unstable wide walk. They often feel tremors in their torso, causing sideways jerks and lunges.[11]
In brain MRI, there is often an increased T2 signalling at the posterior columns of the spinal cord in patients with myelopathy caused by copper deficiency.[4][7][12] T2 signalling is often an indicator of some kind of neurodegeneration. There are some changes in the spinal cord MRI involving the thoracic cord, the cervical cord or sometimes both.[4][7] Copper deficiency myelopathy is often compared to subacute combined degeneration (SCD).[10] Subacute combined degeneration is also a degeneration of the spinal cord, but instead vitamin B12 deficiency is the cause of the spinal degeneration.[4] SCD also has the same high T2 signalling intensities in the posterior column as copper deficient patient in MRI imaging.[12]
Peripheral neuropathy
Another common symptom of copper deficiency is peripheral neuropathy, which is numbness or tingling that can start in the extremities and can sometimes progress radially inward towards the torso.[7][13] In an Advances in Clinical Neuroscience & Rehabilitation (ACNR) published case report, a 69-year-old patient had progressively worsened neurological symptoms.[14] These symptoms included diminished upper limb reflexes with abnormal lower limb reflexes, sensation to light touch and pin prick was diminished above the waist, vibration sensation was lost in the sternum, and markedly reduced proprioception or sensation about the self’s orientation.[14] Many people with the neurological effects of copper deficiency complain about very similar or identical symptoms as the patient.[4][13] This numbness and tingling poses danger for the elderly because it increases their risk of falling and injuring themselves. Peripheral neuropathy can become very disabling leaving some patients dependent on wheelchairs or walking canes for mobility if there is a lack of correct diagnosis. Rarely can copper deficiency cause major disabling symptoms. The deficiency will have to be present for an extensive amount of time until such disabling conditions manifest.
Optic neuropathy
Some patients with copper deficiency have shown signs of vision and color loss.[13] The vision is usually lost in the peripheral views of the eye.[13] The bilateral vision loss is usually very gradual.[13][15] An optical coherence tomography (OCT) shows some nerve fiber layer loss in most patients, suggesting the vision loss and color vision loss was secondary to optic neuropathy or neurodegeneration.[13]
Causes
Surgery
Bariatric surgery is a common cause of copper deficiency.[4][6] Bariatric surgery, such as gastric bypass surgery, is often used for weight control of the morbidly obese. The disruption of the intestines and stomach from the surgery can cause absorption difficulties not only as regards copper but also for iron and vitamin B12 and many other nutrients.[4] The symptoms of copper deficiency myelopathy may take up to decades to develop.
Zinc toxicity
Increased consumption of zinc is another cause of copper deficiency.[7] Zinc is often used for the prevention or treatment of common colds and sinusitis (inflammation of sinuses due to an infection), ulcers, sickle cell disease, celiac disease, memory impairment, and acne.[7] Zinc is found in many common vitamin supplements and is also found in denture creams.[7][15][16] Recently, several cases of copper deficiency myeloneuropathy were found to be caused by prolonged use of denture creams containing high quantities of zinc.[15][16] A literature review found two cases of copper deficiency myelopathy secondary to parenteral zinc supplementation during chronic dialysis.[17]
Metallic zinc is the core of all United States currency coins, including copper-coated pennies. People who ingest a large number of coins will have elevated zinc levels, leading to zinc-toxicity-induced copper deficiency and the associated neurological symptoms. This was the case for a 57-year-old woman diagnosed with schizophrenia. The woman consumed over 600 coins, and started to show neurological symptoms such as unsteady gait and mild ataxia.[18]
Hereditary disorders

Menkes disease is a congenital disease that is a cause of copper deficiency.[3][7][19] Menkes disease is a hereditary condition caused by a defective gene involved with the metabolism of copper in the body.[7] Menkes disease involves a wide variety of symptoms including floppy muscle tone, seizures, abnormally low temperatures, and a peculiar steel color hair that feels very rough.[3][19] Menkes disease is usually a fatal disease with most children dying within the first ten years of life.[3][19]
Other
It is rarely suggested that excess iron supplementation causes copper deficiency myelopathy.[4] Another rarer cause of copper deficiency is celiac disease, probably due to malabsorption in the intestines.[4] Still, a large percentage, around 20%, of cases have unknown causes.[4]
Pathophysiology
Copper functions as a prosthetic group, which permits electron transfers in key enzymatic pathways like the electron transport chain.[4][2][20] Copper is integrated in the enzymes cytochrome c oxidase, which is involved in cellular respiration and oxidative phosphorylation, Cu/Zn dismutase, which is involved in antioxidant defense, and many more listed in the table below.[2]
| Group | Enzyme | Function |
|---|---|---|
| Oxidases | Flavin-containing amine oxidase | Metabolism of neurotransmitters: noradrenaline, dopamine, serotonin and some dietary amines |
| Protein-lysine-6-oxidase (lysyl oxidase) | Connective tissue synthesis- cross-linking of collagen and elastin | |
| Copper-containing amine oxidase (a family of enzymes which includes primary-amine oxidase and diamine oxidase) | Oxidation of biogenic amines including neurotransmitters, histamines, putrescine, cadaverine, and xenobiotic amines | |
| Cytochrome c oxidase | Oxidative phosphorylation, electron transport in the mitochondrial membrane | |
| Superoxide dismutase (Cu/Zn dismutase) | Antioxidant and free radical scavenger, oxidizes dangerous superoxides to safer hydrogen peroxide | |
| Ferroxidase I (ceruloplasmin) | Iron transport-oxidation of Fe2+ to Fe3+, copper storage and transport, antioxidant and free radical neutralizer | |
| Hephaestin (ferroxidase) | Iron transport and oxidation of Fe2+ to Fe3+ in intestinal cells to enable iron uptake | |
| Monooxygenases | Dopamine beta-monooxygenase | Conversion of dopamine to norepinephrine |
| Peptidylglycine monooxygenase | Peptide hormone maturation- amidation of alpha-terminal carboxylic acid group of glycine | |
| Monophenol monooxygenase (Tyrosinase) | Melanin synthesis | |
| Methylation Cycle | Methionine synthase | Transfer of methyl group from methyltetrahydrofolate to homocysteine to generate methionine for the methylation cycle and tetrahydrofolate for purine synthesis |
| Adenosylhomocysteinase (S-Adenosyl-L-homocysteine) | Regeneration of homocysteine from adenosylhomocyesteine (S-Adenosyl-L-homocysteine) in the methylation cycle |
Neurological

Cytochrome c oxidase
There have been several hypotheses about the role of copper and some of its neurological manifestations. Some suggest that disruptions in cytochrome c oxidase, also known as Complex IV, of the electron transport chain is responsible for the spinal cord degeneration.[4][10]
Methylation cycle

Another hypothesis is that copper deficiency myelopathy is caused by disruptions in the methylation cycle.[10] The methylation cycle causes a transfer of a methyl group (-CH3) from methyltetrahydrofolate to a range of macromolecules by the suspected copper dependent enzyme methionine synthase.[10] This cycle is able to produce purines, which are a component of DNA nucleotide bases, and also myelin proteins.[10] The spinal cord is surrounded by a layer of protective protein coating called myelin (see figure). When this methionine synthase enzyme is disrupted, the methylation decreases and myelination of the spinal cord is impaired. This cycle ultimately causes myelopathy.[10]
Hematological cause
Iron transportation
The anemia caused by copper deficiency is thought to be caused by impaired iron transport. Hephaestin is a copper containing ferroxidase enzyme located in the duodenal muscosa that oxidizes iron and facilitates its transfer across the basolateral membrane into circulation.[6] Another iron transporting enzyme is ceruloplasmin.[6] This enzyme is required to mobilize iron from the reticuloendothelial cell to plasma.[6] Ceruloplasmin also oxidizes iron from its ferrous state to the ferric form that is required for iron binding.[3] Impairment in these copper dependent enzymes that transport iron may cause the secondary iron deficiency anemia.[6] Another speculation for the cause of anemia is involving the mitochondrial enzyme cytochrome c oxidase (complex IV in the electron transport chain). Studies have shown that animal models with impaired cytochrome c oxidase failed to synthesize heme from ferric iron at the normal rate.[6] The lower rate of the enzyme might also cause the excess iron to clump, giving the heme an unusual pattern.[6] This unusual pattern is also known as ringed sideroblastic anemia cells.
Cell growth halt
The cause of neutropenia is still unclear; however, the arrest of maturing myelocytes, or neutrophil precursors, may cause the neutrophil deficiency.[2][6]
Zinc intoxication
Zinc intoxication may cause anemia by blocking the absorption of copper from the stomach and duodenum.[4] Zinc also upregulates the expression of chelator metallothionein in enterocytes, which are the majority of cells in the intestinal epithelium.[4] Since copper has a higher affinity for metallothionein than zinc, the copper will remain bound inside the enterocyte, which will be later eliminated through the lumen.[4] This mechanism is exploited therapeutically to achieve negative balance in Wilson’s disease, which involves an excess of copper.[4]
Diagnosis
The diagnosis of copper deficiency may be supported by a person's report of compatible signs and symptoms, findings from a thorough physical examination, and supportive laboratory evidence. Low levels of copper and ceruloplasmin in the serum are consistent with the diagnosis as is a low 24 hour urine copper level.[21] Additional supportive bloodwork findings also include neutropenia and anemia.[21] MRI imaging may demonstrate increased T2 signal of the dorsal column–medial lemniscus pathways.[21]
Treatment
Copper deficiency is a very rare disease and is often misdiagnosed several times by physicians before concluding the deficiency of copper through differential diagnosis (copper serum test and bone marrow biopsy are usually conclusive in diagnosing copper deficiency). On average, patients are diagnosed with copper deficiency around 1.1 years after their first symptoms are reported to a physician.[4] Copper deficiency can be treated with either oral copper supplementation or intravenous copper.[7] If zinc intoxication is present, discontinuation of zinc may be sufficient to restore copper levels back to normal, but this usually is a very slow process.[7] People with zinc intoxication will usually have to take copper supplements in addition to ceasing zinc consumption. Hematological manifestations are often quickly restored back to normal.[7] The progression of the neurological symptoms will be stopped and sometimes improved with appropriate treatment, but residual neurological disability is common.[21]
See also
References
- ↑ Scheiber, Ivo; Dringen, Ralf; Mercer, Julian F. B. (2013). "Chapter 11. Copper: Effects of Deficiency and Overload". In Astrid Sigel, Helmut Sigel and Roland K. O. Sigel (ed.). Interrelations between Essential Metal Ions and Human Diseases. Metal Ions in Life Sciences. Vol. 13. Springer. pp. 359–387. doi:10.1007/978-94-007-7500-8_11. PMID 24470097.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 Halfdanarson, T.R.; Kumar, N.; Li, C.Y.; Phyliky, R.L.; Hogan, W.J. (2008). "Hematological manifestations of copper deficiency: a retrospective review". European Journal of Haematology. 80 (6): 523–531. doi:10.1111/j.1600-0609.2008.01050.x. PMID 18284630.
- ↑ 3.0 3.1 3.2 3.3 3.4 Kodama, H.; Fujisawa, C. (2009). "Copper metabolism and inherited copper transport disorders: molecular mechanisms, screening, and treatment". Metallomics. 1 (1): 42–52. doi:10.1039/B816011M.
- ↑ 4.00 4.01 4.02 4.03 4.04 4.05 4.06 4.07 4.08 4.09 4.10 4.11 4.12 4.13 4.14 4.15 4.16 4.17 4.18 4.19 4.20 4.21 Jaiser, S.R.; Winston, G.P. (2010). "Copper deficiency myelopathy". Journal of Neurology. 257 (6): 869–881. doi:10.1007/s00415-010-5511-x. PMC 3691478. PMID 20232210.
- ↑ "Copper Information: Benefits, Deficiencies, Food Sources". Archived from the original on 2020-11-09. Retrieved 2022-05-04.
- ↑ 6.0 6.1 6.2 6.3 6.4 6.5 6.6 6.7 6.8 6.9 Klevay, L.M. (2006). ""Myelodysplasia," myeloneuropathy, and copper deficiency". Mayo Clinic Proceedings. 81 (1): 132. doi:10.4065/81.1.132. PMID 16438490.
- ↑ 7.00 7.01 7.02 7.03 7.04 7.05 7.06 7.07 7.08 7.09 7.10 7.11 7.12 7.13 7.14 Kumar, N. (2006). "Copper deficiency myelopathy (human swayback)". Mayo Clinic Proceedings. 81 (10): 1371–84. doi:10.4065/81.10.1371. PMID 17036563.
- ↑ 8.0 8.1 Fong, T.; Vij, R.; Vijayan, A.; DiPersio, J.; Blinder, M. (2007). "Copper deficiency: an important consideration in the differential diagnosis of myelodysplastic syndrome". Haematologica. 92 (10): 1429–30. doi:10.3324/haematol.11314. PMID 18024379. S2CID 12958829.
- ↑ Schleper, B.; Stuerenburg, H.J. (2001). "Copper deficiency-associated myelopathy in a 46-year-old woman". Journal of Neurology. 248 (8): 705–6. doi:10.1007/s004150170118. PMID 11569901. S2CID 10318175.
- ↑ 10.0 10.1 10.2 10.3 10.4 10.5 10.6 Jaiser, S.R.; Winston, G.P. (2008). "Copper deficiency myelopathy and subacute combined degeneration of the cord: why is the phenotype so similar?". Journal of Neurology. 255: 569. PMID 18472229.
- ↑ Ataxic gait demonstration. Online Medical Video Archived 2021-05-05 at the Wayback Machine
- ↑ 12.0 12.1 Bolamperti, L.; Leone, M. A.; Stecco, A.; Reggiani, M.; Pirisi, M.; Carriero, A.; et al. (2009). "Myeloneuropathy due to copper deficiency: clinical and MRI findings after copper supplementation". Neurological Sciences. 30 (6): 521–4. doi:10.1007/s10072-009-0126-7. PMID 19768378. S2CID 21488713.
- ↑ 13.0 13.1 13.2 13.3 13.4 13.5 Pineles, S.L.; Wilson, C.A.; Balcer, L.J.; Slater, R.; Galetta, S.L. (2010). "Combined optic neuropathy and myelopathy secondary to copper deficiency". Survey of Ophthalmology. 55 (4): 386–392. doi:10.1016/j.survophthal.2010.02.002. PMID 20451943.
- ↑ 14.0 14.1 Jaiser, S.R.; Duddy, R. (2007). "Copper deficiency masquerading as subacute combined degeneration of the cord and myelodysplastic syndrome" (PDF). ACNR: Advances in Clinical Neuroscience and Rehabilitation. 7 (3): 20–21. Archived (PDF) from the original on 2020-08-01. Retrieved 2022-05-04.
- ↑ 15.0 15.1 15.2 Spinazzi, M.; De Lazzari, F.; Tavolato, B.; Angelini, C.; Manara, R.; Armani, M. (2007). "Myelo-optico-neuropathy in copper deficiency occurring after partial gastrectomy. Do small bowel bacterial overgrowth syndrome and occult zinc ingestion tip the balance?". Journal of Neurology. 254 (8): 1012–7. doi:10.1007/s00415-006-0479-2. PMID 17415508. S2CID 28373986.
- ↑ 16.0 16.1 Hedera, P.; Peltier, A.; Fink, J.K.; Wilcock, S.; London, Z.; Brewer, G.J. (2009). "Myelopolyneuropathy and pancytopenia due to copper deficiency and high zinc levels of unknown origin II. The denture cream is a primary source of excessive zinc". Neurotoxicology. 30 (6): 996–9. doi:10.1016/j.neuro.2009.08.008. PMID 19732792.
- ↑ Rissardo JP, Caprara AL. "Copper deficiency myelopathy secondary to parenteral zinc supplementation during chronic dialysis" (PDF). Neurology Asia. 24: 79–82. Archived (PDF) from the original on 2022-01-20. Retrieved 2022-05-04 – via EBSCO.
- ↑ Dhawan, S.S.; Ryder, K.M.; Pritchard, E. (2008). "Massive penny ingestion: The loot with local and systemic effects". Journal of Emergency Medicine. 35 (1): 33–37. doi:10.1016/j.jemermed.2007.11.023. PMID 18180130.
- ↑ 19.0 19.1 19.2 Kaler, S.G.; Liew, C.J.; Donsante, A.; Hicks, J.D.; Sato, S.; Greenfield, J.C. (2010). "Molecular correlates of epilepsy in early diagnosed and treated Menkes disease". Journal of Inherited Metabolic Disease. 33 (5): 583–9. doi:10.1007/s10545-010-9118-2. PMC 3113468. PMID 20652413.
- ↑ Vest, Katherine E.; Hashemi, Hayaa F.; Cobine, Paul A. (2013). "Chapter 13: The Copper Metallome in Eukaryotic Cells". In Banci, Lucia (ed.). Metallomics and the Cell. Metal Ions in Life Sciences. Vol. 12. Springer. pp. 451–78. doi:10.1007/978-94-007-5561-1_13. ISBN 978-94-007-5560-4. PMID 23595680. electronic-book ISBN 978-94-007-5561-1 ISSN 1559-0836 electronic-ISSN 1868-0402
- ↑ 21.0 21.1 21.2 21.3 Goodman, JC (December 2015). "Neurological Complications of Bariatric Surgery". Current Neurology and Neuroscience Reports. 15 (12): 79. doi:10.1007/s11910-015-0597-2. PMID 26493558. S2CID 21401030.
External links
| Classification |
|---|