Carney complex
| Carney complex | |
|---|---|
| Other names: LAMB syndrome, NAME syndrome | |
Carney complex is an autosomal dominant condition characterized by myxomas of the heart and skin, hyperpigmentation of the skin (lentiginosis), and endocrine overactivity.[1] It is distinct from Carney's triad.
There condition is relatively rare, with less tham 1,000 cases worldwide.[1] Around two-thirds of cases also have have an affected parent.[1] Approximately 7% of all cardiac myxomas are associated with Carney complex.[2]
Symptoms and signs
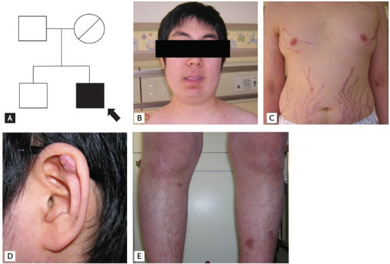
The spotty skin pigmentation and lentigines occur most commonly on the face, especially on the lips, eyelids, conjunctiva and oral mucosa.[3] Cardiac myxomas may lead to embolic strokes and heart failure[2] and may present with fever, joint pain, shortness of breath, diastolic rumble and tumor plop. Myxomas may also occur outside the heart, usually in the skin and breast. Endocrine tumors may manifest as disorders such as Cushing syndrome. The most common endocrine gland manifestation is an ACTH-independent Cushing's syndrome due to primary pigmented nodular adrenocortical disease (PPNAD).[4]
The LAMB acronym refers to lentigines, atrial myxomas, and blue nevi.[5] NAME refers to nevi, atrial myxoma, myxoid neurofibromas, and ephelides.[5]
Testicular cancer, particularly Sertoli cell type, is associated with Carney syndrome.[6] Thyroid and pancreas cancer may also occur.[7][8]
Although J Aidan Carney also described Carney's triad it is entirely different.[9]
Pathophysiology
Carney complex is most commonly caused by mutations in the PRKAR1A gene on chromosome 17 (17q23-q24)[10] which may function as a tumor-suppressor gene. The encoded protein is a type 1A regulatory subunit of protein kinase A. Inactivating germline mutations of this gene are found in 70% of people with Carney complex.[11]
Less commonly, the molecular pathogenesis of Carney complex is a variety of genetic changes at chromosome 2 (2p16).[12][13]
Both types of Carney complex are autosomal dominant. Despite dissimilar genetics, there appears to be no phenotypic difference between PRKAR1A and chromosome 2p16 mutations.[12]
Diagnosis
The diagnosis for this condition is consistent with genetic analysis[14]
Treatment
Cardiac myxomas can be difficult to manage surgically because of recurrence within the heart, often far away from the site of the initial tumor.[3][2]
History
In 1914 an American neurosurgeon, Harvey Cushing, reported on a patient with a pituitary tumour on whom he had operated.[15] The post mortem findings as reported were consistent with Carney complex, though at the time this condition had yet to be described. In 2017 archived tissue from the operation in Cushing's report was subjected to DNA sequencing, revealing an Arg74His (arginine to histidine: guanine (G)-> adenosine (A) transition in the second codon position of the 74th codon in the protein) mutation in the PRKAR1A gene, confirming a diagnosis of Carney complex. Therefore, Cushing's paper appears to be the first report of this complex.[citation needed]
See also
References
- ↑ 1.0 1.1 1.2 WHO Classification of Tumours Editorial Board (2021). "10. Genetic tumour syndromes involving the thorax: Carney complex". Thoracic Tumours. Vol. 5 (5th ed.). Lyon (France): World Health Organization. pp. 478–479. ISBN 978-92-832-4506-3. Archived from the original on 2022-05-14. Retrieved 2022-07-26.
- ↑ 2.0 2.1 2.2 Reynen, K. (1995). "Cardiac Myxomas". New England Journal of Medicine. 333 (24): 1610–1617. doi:10.1056/NEJM199512143332407. PMID 7477198.
- ↑ 3.0 3.1 McCarthy, P.; Piehler, J.; Schaff, H.; Pluth, J.; Orszulak, T.; Vidaillet Jr, H.; Carney, J. (1986). "The significance of multiple, recurrent, and "complex" cardiac myxomas". The Journal of Thoracic and Cardiovascular Surgery. 91 (3): 389–396. doi:10.1016/s0022-5223(19)36054-4. PMID 3951243.
- ↑ "Carney Complex". National Center for Biotechnology Information, U.S. National Library of Medicine. Archived from the original on 28 August 2021. Retrieved 5 July 2021.
- ↑ 5.0 5.1 Carney Syndrome at eMedicine
- ↑ Campbell Walsh urology, 10th edition, page 1693
- ↑ Gaujoux S, Tissier F, Ragazzon B, Rebours V, Saloustros E, Perlemoine K, Vincent-Dejean C, Meurette G, Cassagnau E, Dousset B, Bertagna X, Horvath A, Terris B, Carney JA, Stratakis CA, Bertherat J (2011). "Pancreatic ductal and acinar cell neoplasms in Carney complex: a possible new association". J Clin Endocrinol Metab. 96 (11): E1888–95. doi:10.1210/jc.2011-1433. PMC 3205895. PMID 21900385.
- ↑ Bano G, Hodgson S (2016). "Diagnosis and Management of Hereditary Thyroid Cancer". Recent Results Cancer Res. Recent Results in Cancer Research. 205: 29–44. doi:10.1007/978-3-319-29998-3_3. ISBN 978-3-319-29996-9. PMID 27075347.
- ↑ Gaissmaier C (December 1999). "Carney complex" (letter and response). Circulation. 100 (25): e150. doi:10.1161/01.cir.100.25.e150. PMID 10604916. Archived from the original on 2006-06-27. Retrieved 2021-07-06.
- ↑ Online Mendelian Inheritance in Man (OMIM): Carney Complex, type 1; CNC1 - 160980
- ↑ "Cardiac Myxoma". The Lecturio Medical Concept Library. Archived from the original on 9 July 2021. Retrieved 6 July 2021.
- ↑ 12.0 12.1 Stratakis, C. A.; Kirschner, L. S.; Carney, J. A. (2001). "Clinical and Molecular Features of the Carney Complex: Diagnostic Criteria and Recommendations for Patient Evaluation". Journal of Clinical Endocrinology & Metabolism. 86 (9): 4041–4046. doi:10.1210/jc.86.9.4041. PMID 11549623.
- ↑ Online Mendelian Inheritance in Man (OMIM): Carney Complex, type 2; CNC2 - 605244
- ↑ RESERVED, INSERM US14-- ALL RIGHTS. "Orphanet: Carney complex". www.orpha.net. Archived from the original on 17 January 2021. Retrieved 19 August 2021.
- ↑ Tsay CJ, Stratakis CA, Faucz FR, London E, Stathopoulou C, Allgauer M, Quezado M, Dagradi T, Spencer DD, Lodish M (2017) Harvey Cushing treated the first known patient with Carney complex. J Endocr Soc 1(10):1312-1321. doi: 10.1210/js.2017-00283
External links
| Classification | |
|---|---|
| External resources |
- GeneReview/UW/NIH entry on Carney complex Archived 2010-06-05 at the Wayback Machine
- Pages with script errors
- Articles with hatnote templates targeting a nonexistent page
- All articles with unsourced statements
- Articles with unsourced statements from March 2021
- Articles with invalid date parameter in template
- Webarchive template wayback links
- Hereditary cancers
- Cardiology
- Melanocytic nevi and neoplasms
- Syndromes affecting the skin
- Syndromes affecting the heart
- Syndromes with tumors