Blastomyces dermatitidis
| Blastomyces dermatitidis | |
|---|---|
| |
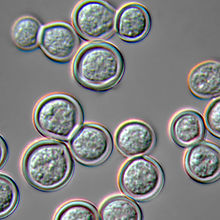
| Blastomyces dermatitidis, yeast form | |
| Scientific classification | |
| Kingdom: | |
| Division: | |
| Class: | |
| Order: | |
| Family: | |
| Genus: | |
| Species: | B. dermatitidis
|
| Binomial name | |
| Blastomyces dermatitidis Gilchrist & W.R.Stokes (1898)
| |
Blastomyces dermatitidis is a dimorphic fungus that causes blastomycosis, an invasive and often serious fungal infection found occasionally in humans and other animals.[1] It lives in soil and wet, decaying wood, often in an area close to a waterway such as a lake, river or stream.[1] Indoor growth may also occur, for example, in accumulated debris in damp sheds or shacks. The fungus is endemic to parts of eastern North America, particularly boreal northern Ontario, southeastern Manitoba, Quebec south of the St. Lawrence River, parts of the U.S. Appalachian mountains and interconnected eastern mountain chains, the west bank of Lake Michigan, the state of Wisconsin, and the entire Mississippi Valley including the valleys of some major tributaries such as the Ohio River. In addition, it occurs rarely in Africa both north and south of the Sahara Desert, as well as in the Arabian Peninsula and the Indian subcontinent. Though it has never been directly observed growing in nature, it is thought to grow there as a cottony white mold, similar to the growth seen in artificial culture at 25 °C (77 °F). In an infected human or animal, however, it converts in growth form and becomes a large-celled budding yeast.[1] Blastomycosis is generally readily treatable with systemic antifungal drugs once it is correctly diagnosed; however, delayed diagnosis is very common except in highly endemic areas.
Morphology and phylogeny
Blastomyces dermatitidis is the causal agent of blastomycosis, a potentially very serious disease that typically begins with a characteristically subtle pneumonia-like infection that may progress, after 1–6 months, to a disseminated phase that causes lesions to form in capillary beds throughout the body, most notably the skin, internal organs, central nervous system and bone marrow. The sexual form of this fungus was formerly known as Ajellomyces dermatitidis.[2]
In 2013, a second species was described in the genus Blastomyces, B. gilchristii, which subsumes certain strains previously assigned to B. dermatitidis.[3] Three more species have been described: Blastomyces emzantsi, Blastomyces parvus and Blastomyces percursus.
Despite widespread use, the genus Blastomyces is currently invalid under the International Code of Botanical Nomenclature.[4] This is because under Article 53.1 of the Code, a taxon name is illegitimate if it "is spelled exactly like a name based on a different type that was previously and validly published for a taxon at the same rank",[5] and the name Blastomyces had previously been published for the fungus now known as Chrysosporium.[6]
Along with two other important human-pathogenic fungi, Histoplasma capsulatum, Paracoccidioides brasiliensis and Polytolypa hystricis, species of Blastomyces belong to the family Ajellomycetaceae.[7] The three principal pathogens in this family are all grouped physiologically as "dimorphic fungi": fungi that switch from a mold-like (filamentous) growth form in the natural habitat to a yeast-like growth form in the warm-blooded animal host. Blastomyces dermatitidis itself is a sexual organism, occurring in nature as both a + mating type and a − mating type. This is epidemiologically important for two reasons: firstly, it implies that the organism will be genetically variable, potentially leading to variations in disease severity, treatment response and habitat preference; secondly, it implies that a suitable, stable habitat must exist for the complex process of sexual reproduction to take place. This habitat is as yet unknown. In its asexual form, the fungus grows as a typical colonial microfungus, comparable to Penicillium or Rhizopus mold forms commonly seen on mouldy bread.
In nature, the fungus forms a network of thread-like mycelium that penetrates the substratum on which it grows, and then after 3–5 days of growth begins to reproduce asexually with small (2–10 µm) conidia (asexual spores). These conidia are probably the main infectious particles produced by the fungus. They form on individual short stalks and readily become airborne when the colony is disturbed; their size places them well within the respirable size range for particles,[8] meaning that they can deposit deeply in the lungs when inhaled. Sexual reproduction by the fungus requires the meeting of colonies of + and – mating type, probably a relatively rare event, and results in the production of small ascomata (sexual fruiting bodies) 200–350 µm, looking, to the naked eye, similar to a woollen fuzz ball, and in microscopic view consisting of a layer of spiralling, springy guard hairs surrounding a fertile core in which groups of 8 ascospores (sexual spores) are produced in small round reproductive sacs (asci). The ascospores, at 1.5–2.0 µm, are among the smallest reproductive particles produced by fungi, and are within the respirable size range.[8] The budding yeast cells seen in infected tissues and bodily fluids are generally relatively large (c. 8–15 µm) and characteristically bud through a broad base or neck, making them highly recognizable to the pathologist. A small ("nanic") form is rarely seen with cells under 6 µm.
Geographic distribution and variants
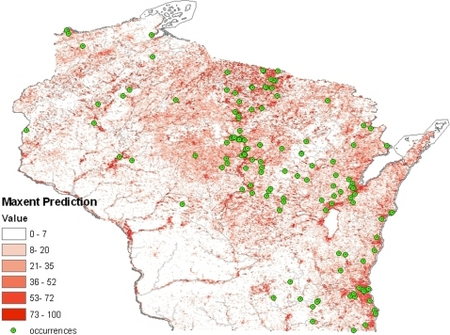
One of the unexplained regularities of nature is that there are several fungi of different phylogenetic ancestry that show a similar pattern of existence: dimorphism (conversion from a filamentous form in the environment to a yeast form in warm-blooded host tissues), virulent pathogenesis (ability to cause a significant infection in an animal host that is otherwise in good health), pulmonary infectivity (infection mainly via the lungs) and sharply delimited endemism (occurrence in only a limited geographic range.). Blastomyces dermatitidis is one of these fungi; the others are Histoplasma capsulatum, Paracoccidioides brasiliensis, Coccidioides immitis, C. posadasii and Talaromyces marneffei.
The geographic range of B. dermatitidis is largely focused around the waterways of the St. Lawrence and Mississippi River systems of North America. There is a widely distributed and much republished, partially erroneous map that shows the U.S. portion of this range accurately, inclusive of occurrence in Minnesota, Wisconsin, Ohio, Kentucky, Arkansas, Tennessee, North and South Carolina, the Virginias, Mississippi, Louisiana, and a few regions of states adjacent to those named.[9] The Canadian range of B. dermatitidis shows an abundance of blastomycosis in broad areas north and south of the St. Lawrence River in Quebec, as well as high endemicity along the north shore of Lake Erie and the low endemicity in southeastern corner of Manitoba. Though the Quebec distribution is reasonably accurate, the rest of Canada is strongly misrepresented. Blastomyces dermatitidis is absent or nearly so from the Lake Erie area, but occurs sporadically on the north shore of Lake Ontario, including metropolitan Toronto,[10] and, most notably, has areas of high endemicity throughout northern Ontario.[11] Remarkably high incidence is noted for some parts of the Kenora area and climatologically similar areas of northwestern Ontario.[12] To the west, the range of endemic blastomycosis extends across southern Manitoba and into adjacent Saskatchewan.[13] A few cases have been reported from north central Alberta, e.g., the Edmonton area, though in these cases an atypical genetic group of the fungus may be involved.[14]
In the rest of the world, B. dermatitidis occurs at low levels in various parts of Africa, from Algeria to South Africa, as well as in and near the Arabian Peninsula. The African isolates are divided into two biologically different antigen groups: isolates from north of the Sahara are similar to North American isolates in having A and K antigens, while southern African isolates lack the A antigen.[15] Isolates from the middle east possess both antigens. The sub-Saharan African isolates differ in the laboratory from other isolates by being exceedingly difficult to convert to the yeast phase, and they also show some enzymatic distinctions.[16]
Ecology
Blastomyces dermatitidis is one of the most ecologically mysterious organisms causing human and animal disease. Prediction of disease risk and prevention of disease are both made extraordinarily difficult by our very poor understanding of where and how this organism normally grows in nature. Despite decades of attempts at isolating organisms from epidemiological foci, B. dermatitidis has only been isolated from the environment 21 times.[17] Most of these isolations have been based on the arduous isolation techniques involving the suspension of soil or other environmental materials in aqueous medium with antibacterial antibiotics, and injection of mice with these materials, followed by sacrifice of the animals when they appear ill or at the end of six weeks.[18] The internal organs of the mice are then checked microscopically for evidence of blastomycosis. Needless to say, the cost and complexity of performing such studies is imposing, especially as the ethical clearance procedures for work involving animals become ever more involved. More direct and economical mycological techniques for environmental isolation, such as dilution plating, have never yielded positive results for Blastomyces growth. Since B. dermatitidis will grow readily from clinical samples on common laboratory media, the lack of success in isolating it from environmental materials is generally ascribed to the inhibitory effects of co-occurring common molds and antibiotic-resistant bacteria.
In just one experiment, a single positive B. dermatitidis culture was gained via use of a novel enrichment broth technique.[19] Recently, in an important breakthrough, a specific PCR technique was developed that was able to detect B. dermatitidis in three environmental samples from a dog kennel that had been experiencing problems with blastomycosis.[17]
What has been learned from direct isolation and recent PCR studies is that B. dermatitidis tends to be associated with soils and wood debris in areas “characterized by an acidic pH, high organic content (due to rotting or decayed wood or vegetation and animal or bird droppings), abundant moisture, and proximity to waterways”.[20] Recent PCR detections, for example, concerned a Kentucky dog kennel where 35 of 100 dogs had contracted blastomycosis.[17] Previous isolations have been from comparable sites such as soil and wood debris from an abandoned Wisconsin beaver dam,[21] and woody materials from a Wisconsin woodpile.[19] Isolation of B. dermatitidis was also accomplished from an earthen floor indoors on one occasion.[22]
There has been a long history of justifiable speculation that B. dermatitidis may associate in nature with one or more indigenous North American mammalian host species. To date, however, all the animal species that have been subjected to focused investigation have been exonerated of this specific connection. Unsubstantiated suspicion has particularly focused on the beaver,[21][23][24] but the shrew,[25] the bat[26] and the prairie dog[27] have also been focal points of interest, with no conclusive interspecies association being demonstrated to date. The closely related pathogenic fungus P. brasiliensis in South America has a well substantiated, though not well understood, ecological link with the nine-banded armadillo, Dasypus novemcinctus.[28] This member of the mammalian order Edentata has no close relatives in the geographic range of B. dermatitidis.
Disease
Avoidance of exposure in endemic areas is the principal means of disease prevention. Because the agent is known to distribute in dusts, the minimization of dust-generating activities, such as digging, sweeping, etc., is key. Although a method of soil decontamination has been described and demonstrated to be effective, it uses hazardous chemicals and its use is best reserved for situations that cannot be managed otherwise.[18]
References
- ↑ 1.0 1.1 1.2 DiSalvo, A.F. (1992). Al-Doory, Y.; DiSalvo, A.F. (eds.). Ecology of Blastomyces dermatitidis. Plenum. pp. 43–73.
- ↑ Bennett, John E. (2014). "Introduction to Mycoses". In John E. Bennett; Raphael Dolin; Martin J. Blaser (eds.). Mandell, Douglas, and Bennett's Principles and Practice of Infectious Diseases. Elsevier Health Sciences. p. 2874.
- ↑ Brown, Elizabeth M.; McTaggart, Lisa R.; Zhang, Sean X.; Low, Donald E.; Stevens, David A.; Richardson, Susan E.; Litvintseva, Anastasia P. (22 March 2013). "Phylogenetic Analysis Reveals a Cryptic Species Blastomyces gilchristii, sp. nov. within the Human Pathogenic Fungus Blastomyces dermatitidis". PLOS ONE. 8 (3): e59237. Bibcode:2013PLoSO...859237B. doi:10.1371/journal.pone.0059237. PMC 3606480. PMID 23533607.
- ↑ Peterson, SW; Sigler, L (1998). "Molecular genetic variation in Emmonsia crescens and Emmonsia parva, etiologic agents of adiaspiromycosis, and their phylogenetic relationship to Blastomyces dermatitidis (Ajellomyces dermatitidis) and other systemic fungal pathogens". Journal of Clinical Microbiology. 36 (10): 2918–25. doi:10.1128/JCM.36.10.2918-2925.1998. PMC 105088. PMID 9738044.
- ↑ "Chapter VIII: Rejection of Names: Article 53". International Code of Nomenclature for algae, fungi, and plants (Shenzhen Code). 2018. Archived from the original on 2022-06-01. Retrieved 2022-05-14.
- ↑ "Index Fungorum - Names Record". Index Fungorum. Archived from the original on 2022-05-15. Retrieved 2022-05-14.
- ↑ Untereiner, Wendy A.; Scott, James A.; Naveau, F.; Sigler, Lynne; Bachewich, J.; Angus, A. (2004). "The Ajellomycetaceae, a new family of vertebrate-associated Onygenales". Mycologia. 96 (4): 812–21. doi:10.2307/3762114. JSTOR 3762114. PMID 21148901. Archived from the original on 2011-09-30. Retrieved 2022-11-23.
- ↑ 8.0 8.1 Lippman, M (2001). Size-selective health hazard sampling. Pp. . In BS Cohen, CS McCammon (eds.), Air Sampling Instruments, 9th edition. ACGIH Press, Cincinnati, Ohio. Cincinnati, Ohio: in BS Cohen, CS McCammon (eds.), Air Sampling Instruments, 9th edition. ACGIH Press. pp. 93–134.
- ↑ Kwon-Chung, K.J., Bennett, J.E.; Bennett, John E. (1992). Medical Mycology. Philadelphia: Lea & Febiger. ISBN 978-0812114638.
- ↑ Lester, RS; DeKoven, JG; Kane, J; Simor, AE; Krajden, S; Summerbell, RC (2000). "Novel cases of blastomycosis acquired in Toronto, Ontario". Canadian Medical Association Journal. 163 (10): 1309–12. PMC 80342. PMID 11107469.
- ↑ Kane, J; Righter, J; Krajden, S; Lester, RS (1983). "Blastomycosis: a new endemic focus in Canada". Canadian Medical Association Journal. 129 (7): 728–31. PMC 1875443. PMID 6616383.
- ↑ Dwight, P.J.; Naus, M; Sarsfield, P; Limerick, B (2000). "An outbreak of human blastomycosis: the epidemiology of blastomycosis in the Kenora catchment region of Ontario, Canada". Canada Communicable Disease Report. 26 (10): 82–91. PMID 10893821.
- ↑ Vallabh, V; Martin, T; Conly, JM (1988). "Blastomycosis in Saskatchewan". The Western Journal of Medicine. 148 (4): 460–2. PMC 1026149. PMID 3388850.
- ↑ Sekhon, AS; Jackson, FL; Jacobs, HJ (1982). "Blastomycosis: report of the first case from Alberta Canada". Mycopathologia. 79 (2): 65–9. doi:10.1007/bf00468081. PMID 6813742. S2CID 27296444.
- ↑ Frean, JA; Carman, WF; Crewe-Brown, HH; Culligan, GA; Young, CN (1989). "Blastomyces dermatitidis infections in the RSA". South African Medical Journal = Suid-Afrikaanse Tydskrif vir Geneeskunde. 76 (1): 13–6. PMID 2662433.
- ↑ Summerbell, RC; Kane, J; Pincus, DH (1990). "Enzymatic activity profiling as a potential biotyping method for Ajellomyces dermatitidis". Journal of Clinical Microbiology. 28 (5): 1054–6. doi:10.1128/jcm.28.5.1054-1056.1990. PMC 267864. PMID 2351722.
- ↑ 17.0 17.1 17.2 Burgess, JW; Schwan, WR; Volk, TJ (2006). "PCR-based detection of DNA from the human pathogen Blastomyces dermatitidis from natural soil samples". Medical Mycology. 44 (8): 741–8. doi:10.1080/13693780600954749. PMID 17127631.
- ↑ 18.0 18.1 Ajello, L., and R. J. Weeks. 1983. Soil decontamination and other control measures. Pp. 229-238. In A. F. DiSalvo (ed.), Occupational Mycoses. Lea and Febiger, Philadelphia, Pennsylvania.
- ↑ 19.0 19.1 Baumgardner, DJ; Paretsky, DP (1999). "The in vitro isolation of Blastomyces dermatitidis from a woodpile in north central Wisconsin, USA". Medical Mycology. 37 (3): 163–8. doi:10.1111/j.1365-280x.1999.00214.x. PMID 10421847.
- ↑ Baumgardner, DJ; Buggy, BP; Mattson, BJ; Burdick, JS; Ludwig, D (1992). "Epidemiology of blastomycosis in a region of high endemicity in north central Wisconsin". Clinical Infectious Diseases. 15 (4): 629–35. doi:10.1093/clind/15.4.629. PMID 1420675.
- ↑ 21.0 21.1 Klein, Bruce S.; Vergeront, James M.; Weeks, Robert J.; Kumar, U. Nanda; Mathai, George; Varkey, Basil; Kaufman, Leo; Bradsher, Robert W.; Stoebig, James F.; Davis, Jeffrey P. (1986). "Isolation of Blastomyces dermatitidis in soil associated with a large outbreak of blastomycosis in Wisconsin". New England Journal of Medicine. 314 (9): 529–534. doi:10.1056/NEJM198602273140901. PMID 3945290.
- ↑ Bakerspigel, A; Kane, J; Schaus, D (1986). "Isolation of Blastomyces dermatitidis from an earthen floor in southwestern Ontario, Canada". Journal of Clinical Microbiology. 24 (5): 890–1. doi:10.1128/jcm.24.5.890-891.1986. PMC 269057. PMID 3771778.
- ↑ Bradsher, RW (1987). "Water and blastomycosis: don't blame beaver". The American Review of Respiratory Disease. 136 (6): 1324–6. doi:10.1164/ajrccm/136.6.1324. PMID 3688633.
- ↑ Gaus, DP; Baumgardner, DJ; Paretsky, D (1996). "Attempted isolation of Blastomyces dermatitidis from rectal cultures of beaver (Castor canadensis) from north central Wisconsin". Wilderness & Environmental Medicine. 7 (2): 192. doi:10.1580/1080-6032(1996)007[0192:ltte]2.3.co;2. PMID 11990111.
- ↑ Baumgardner, DJ; Summerbell, R; Krajden, S; Alexopoulou, I; Agrawal, B; Bergeson, M; Fuksa, M; Bemis, C; Baumgardner, MA (2005). "Attempted isolation of Blastomyces dermatitidis from native shrews in northern Wisconsin, USA". Medical Mycology. 43 (5): 413–6. doi:10.1080/13693780400008191. PMID 16178369.
- ↑ Chaturvedi, VP; Randhawa, HS; Kini, S; Khan, ZU (1986). "Survival of Blastomyces dermatitidis in the gastrointestinal tract of an orally infected insectivorous bat, Rhinopoma hardwickei hardwickei Gray". Journal of Medical and Veterinary Mycology. 24 (4): 349–52. doi:10.1080/02681218680000521. PMID 3746588.
- ↑ De Groote, MA; Bjerke, R; Smith, H; Rhodes III, LV (2000). "Expanding epidemiology of blastomycosis: clinical features and investigation of 2 cases in Colorado". Clinical Infectious Diseases. 30 (3): 582–4. doi:10.1086/313717. PMID 10722448.
- ↑ Vergara, ML; Martinez, R (1999). "Role of the armadillo Dasypus novemcinctus in the epidemiology of paracoccidioidomycosis". Mycopathologia. 144 (3): 131–3. doi:10.1023/A:1007034215003. PMID 10531678. S2CID 35693829.
External links
- Blastomyces at the US National Library of Medicine Medical Subject Headings (MeSH)
- "Blastomyces dermatitidis". Mycology Online § Dimorphic Fungal Pathogens. University of Adelaide. Archived from the original on 2007-08-17. Retrieved 2007-08-04.
- http://pathmicro.med.sc.edu/mycology/mycology-6.htm Archived 2007-08-18 at the Wayback Machine