Aortic insufficiency
Aortic insufficiency (AI), also known as aortic regurgitation (AR), is a type of valvular heart disease in which there is backward flow of blood from the aorta, through the aortic valve, into the left ventricle, when the heart relaxes.[1] When of sudden onset, symptoms may include a rapid heart rate, cough, shortness of breath, and chest pain.[1] Cases of gradual onset may have few symptoms, though may develop shortness of breath and tiredness.[2] Complications may include heart failure.[2]
Cases of sudden onset may occur due to infective endocarditis, rupture of the ascending aorta with aortic cusp prolapse, and complications of percutaneous aortic balloon valvuloplasty or a prosthetic aortic valve.[1] Cases of gradual onset may occur due to rheumatic heart disease, infective endocarditis, valve degeneration, congenital valve abnormalities such as a bicuspid aortic valve, age related valvular calcification, dilatation of the aorta, aortic dissection, aortitis due to syphilis or giant cell arteritis, hypertension, drug-induced valvulopathy, and osteogenesis imperfecta.[1] Diagnosis may be suspected based on a diastolic murmur and confirmed by echocardiography.[1]
Treatment of cases of sudden onset depends on the underlying cause; however, many cases require urgent surgery.[1] Treatment of cases of gradual onset may include ACE inhibitors, calcium channel blockers, or beta blockers initially.[1] More severe cases may require aortic valve replacement or aortic valve repair.[1] Chronic disease is generally gradual in onset, with follow up recommended at intervals of 6 months to 5 years depending on the current severity.[2]
Aortic insufficiency is common.[2] In adults in the United States about 5% are affected to some degree and about 0.5% are severely affected.[1] It becomes more common with age.[1] It was first described in 1832 by Dominic Corrigan.[1][3]
Signs and symptoms
Symptoms of aortic insufficiency are similar to those of heart failure and include the following:[4][5]
- Dyspnea on exertion
- Orthopnea (shortness of breath when lying flat)
- Paroxysmal nocturnal dyspnea (shortness of breath at night)
- Palpitations
- Heart related chest pain
- Cyanosis (in acute cases)
Causes
In terms of the cause of aortic insufficiency, is often due to the aortic root dilation (annuloaortic ectasia), which is idiopathic in over 80% of cases, but otherwise may result from aging, syphilitic aortitis, osteogenesis imperfecta, aortic dissection, Behçet's disease, reactive arthritis and systemic hypertension.[4] Aortic root dilation is the most common cause of aortic insufficiency in developed countries.[6] Additionally, aortic insufficiency has been linked to the use of some medications, specifically medications containing fenfluramine or dexfenfluramine isomers and dopamine agonists.[7][8] Other potential causes that affect the valve directly include Marfan syndrome, Ehlers–Danlos syndrome, ankylosing spondylitis, and systemic lupus erythematosus. In acute cases of aortic insufficiency, the main causes are infective endocarditis, aortic dissection or trauma.[4]
Pathophysiology

The mechanism of aortic insufficiency (AI), comprises the pressure in the left ventricle falling below the pressure in the aorta, the aortic valve is not able to completely close. This causes a leaking of blood from the aorta into the left ventricle. This means that some of the blood that was already ejected from the heart is regurgitating back into the heart. The percentage of blood that regurgitates back through the aortic valve due to AI is known as the regurgitant fraction. This regurgitant flow causes a decrease in the diastolic blood pressure in the aorta, and therefore an increase in the pulse pressure. Since some of the blood that is ejected during systole regurgitates back into the left ventricle during diastole, there is decreased effective forward flow in AI.[9][10]
While diastolic blood pressure is diminished and the pulse pressure widens, systolic blood pressure generally remains normal or can even be slightly elevated, this is because sympathetic nervous system and the renin-angiotensin-aldosterone axis of the kidneys compensate for the decreased cardiac output.[11] Catecholamines will increase the heart rate and increase the strength of ventricular contraction, directly increasing cardiac output. Catecholamines will also cause peripheral vasoconstriction, which causes increased systemic vascular resistance and ensures that organs are adequately perfused.[12] Renin, a proteolytic enzyme, cleaves angiotensinogen to angiotensin I, which is converted to angiotensin II.[13] In the case of chronic aortic insufficiency with resultant cardiac remodeling, heart failure will develop, and it is possible to see systolic pressures diminish.[14] Aortic insufficiency causes both volume overload (elevated preload) and pressure overload (elevated afterload) of the heart.[15]
The volume overload, due to elevated pulse pressure and the systemic effects of neuroendocrine hormones causes left ventricular hypertrophy (LVH).[10] There is both concentric hypertrophy and eccentric hypertrophy in AI. The concentric hypertrophy is due to the increased left ventricular pressure overload associated with AI, while the eccentric hypertrophy is due to volume overload caused by the regurgitant fraction.[16]
Physiologically, in individuals with a normally functioning aortic valve, the valve is only open when the pressure in the left ventricle is higher than the pressure in the aorta. This allows the blood to be ejected from the left ventricle into the aorta during ventricular systole. The amount of blood that is ejected by the heart is known as the stroke volume. Under normal conditions, >50% of the blood in a filled left ventricle is ejected into the aorta to be used by the body. After ventricular systole, the pressure in the left ventricle decreases as it relaxes and begins to fill up with blood from the left atrium. This relaxation of the left ventricle (early ventricular diastole) causes a fall in its pressure. When the pressure in the left ventricle falls below the pressure in the aorta, the aortic valve will close, preventing blood in the aorta from going back into the left ventricle.[17][18][19]
Diagnosis
In terms of the diagnosis of aortic regurgitation a common test for the evaluation of the severity is transthoracic echocardiography, which can provide two-dimensional views of the regurgitant jet, allow measurement of velocity, and estimate jet volume.[21] The findings in severe aortic regurgitation, based on the 2012 American College of Cardiology/American Heart Association guidelines include:[22][23]
- An AI color jet width > 65 % of the left ventricular outflow tract diameter
- Doppler vena contracta width > 0.6 cm
- The pressure half-time of the regurgitant jet is < 200 ms
- Early termination of the mitral inflow
- Holodiastolic flow reversal in the descending aorta.
- Regurgitant volume > 60 ml
- Regurgitant fraction > 50 %
- Estimated regurgitant orifice area > 0.3 cm2
- Increased left ventricular size
Chest X-ray can assist in making the diagnosis, showing left ventricular hypertrophy and dilated aorta. ECG typically indicates left ventricular hypertrophy. Cardiac chamber catheterization assists in assessing the severity of regurgitation and any left ventricular dysfunction.[4]
Physical examination

The physical examination of an individual with aortic insufficiency involves auscultation of the heart to listen for the murmur of aortic insufficiency and the S3 heart sound (S3 gallop correlates with development of LV dysfunction).[4] The murmur of chronic aortic insufficiency is typically described as early diastolic and decrescendo, which is best heard in the third left intercostal space and may radiate along the left sternal border.[24]
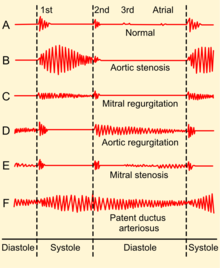
If there is increased stroke volume of the left ventricle due to volume overload, an ejection systolic 'flow' murmur may also be present when auscultating the same aortic area. Unless there is concomitant aortic valve stenosis, the murmur should not start with an ejection click. There may also be an Austin Flint murmur,[4] a soft mid-diastolic rumble heard at the apical area; it appears when a regurgitant jet of blood from severe aortic insufficiency partially closes the anterior mitral leaflet. Peripheral physical signs of aortic insufficiency are related to the high pulse pressure and the rapid decrease in blood pressure during diastole due to blood returning to the heart from the aorta through the incompetent aortic valve, although the usefulness of some of the eponymous signs has been questioned:[25] Phonocardiograms detect AI by having electric voltage mimic the sounds the heart makes.[26]
Characteristics- indicative of aortic regurgitation are as follow:
- Corrigan's pulse [27]
- De Musset's sign [28]
- Quincke sign [28]
- Traube sign [29]
- Duroziez's sign [28]
- Landolfi's sign [29]
- Becker's sign [29]
- Müller's sign [28]
- Mayne's sign [29]
- Rosenbach's sign [29]
- Gerhardt's sign [29]
- Hill's sign [29]
- Lincoln sign [29]
- Sherman sign [29]
Classification
The hemodynamic sequelae of AI are dependent on the rate of onset of AI.[30] Therefore, can be acute or chronic as follows:
- Acute aortic insufficiency In acute AI, as may be seen with acute perforation of the aortic valve due to endocarditis, there will be a sudden increase in the volume of blood in the left ventricle. The ventricle is unable to deal with the sudden change in volume.[31] The filling pressure of the left ventricle will increase. This causes pressure in the left atrium to rise, and the individual will develop pulmonary edema. Severe acute aortic insufficiency is considered a medical emergency. There is a high mortality rate if the individual does not undergo immediate surgery for aortic valve replacement.[10]
- Acute AI usually presents as florid congestive heart failure, and will not have any of the signs associated with chronic AI since the left ventricle had not yet developed the eccentric hypertrophy and dilatation that allow an increased stroke volume, which in turn cause bounding peripheral pulses. On auscultation, there may be a short diastolic murmur and a soft S1. S1 is soft because the elevated filling pressures close the mitral valve in diastole.[medical citation needed]
- Chronic aortic insufficiency If the individual survives the initial hemodynamic derailment that acute AI presents, the left ventricle adapts by its eccentric hypertrophy and dilatation with a subsequent compensated volume overload. The left ventricular filling pressures will revert to normal and the individual will no longer have overt heart failure. In this compensated phase, the individual may be totally asymptomatic and may have normal exercise tolerance. Eventually (typically after a latency period) the left ventricle will become decompensated, and filling pressures will increase. Some individuals enter this decompensated phase asymptomatically, treatment for AI involves aortic valve replacement prior to this decompensation phase.[32]
Treatment
Aortic insufficiency or aortic regurgitation can be treated either medically or surgically, depending on the acuteness of presentation, the symptoms and signs associated with the disease process, and the degree of left ventricular dysfunction.[6][33] Surgical treatment in asymptomatic patients has been recommended if the ejection fraction falls to 50% or below, in the face of progressive and severe left ventricular dilatation, or with symptoms or abnormal response to exercise testing. For both groups of patients, surgery before the development of worsening ejection fraction/LV dilatation is expected to reduce the risk of sudden death, and is associated with lower peri-operative mortality. Also, surgery is optimally performed immediately in acute cases.[4][6]
Medical treatment

Medical therapy of chronic aortic insufficiency that is stable and asymptomatic involves the use of vasodilators.[4] Trials have shown a short term benefit in the use of ACE inhibitors or angiotensin II receptor antagonists, nifedipine, and hydralazine in improving left ventricular wall stress, ejection fraction, and mass.[6] The goal in using these pharmacologic agents is to decrease the afterload so that the left ventricle is somewhat spared.[34] The regurgitant fraction may not change significantly, since the gradient between the aortic and left ventricular pressures is usually fairly low at the initiation of treatment. Other rather conservative medical treatments for stable and asymptomatic cases include low sodium diet, diuretics, digoxin, calcium blockers and avoiding very strenuous activity.[4]
As of 2007, the American Heart Association no longer recommends antibiotics for endocarditis prophylaxis before certain procedures in patients with aortic insufficiency.[35] Antibiotic prophylaxis to prevent endocarditis before gastrointestinal or genitourinary procedures is no longer recommended for any patient with valvular disease.[35] Cardiac stress test is useful in identifying individuals that may be best suited for surgical intervention.[36] Radionuclide angiography is recommended and useful when the systolic wall stress is calculated and combined to the results.[37]
Surgery
| Symptoms | Ejection fraction | Additional Findings |
|---|---|---|
| Present (NYHA II-IV) |
Any | none |
| Absent | > 50% | Abnormal exercise test, severe LV dilatation (systolic ventricular diameter >55 mm) |
| Absent | <=50 % | none |
| Cardiac surgery for other cause (i.e.: CAD, other valvular disease, ascending aortic aneurysm) | ||
A surgical treatment for AI is aortic valve replacement;[38] this is currently an open-heart procedure. In the case of severe acute aortic insufficiency, all individuals should undergo surgery, if there are no absolute contraindications (for surgery).[6][39] Individuals with bacteremia with aortic valve endocarditis should not wait for treatment with antibiotics to take effect, given the high mortality associated with the acute AI. Replacement with an aortic valve homograft should be performed if feasible.[40][41]
Prognosis
The risk of death in individuals with aortic insufficiency, dilated ventricle, normal ejection fraction who are asymptomatic is about 0.2 percent per year. Risk increases if the ejection fraction decreases or if the individual develops symptoms.[39]
Individuals with chronic (severe) aortic regurgitation follow a course that once symptoms appear, surgical intervention is needed. AI is fatal in 10 to 20% of individuals who do not undergo surgery for this condition. Left ventricle dysfunction determines to an extent the outlook for severity of aortic regurgitation cases.[6][42]
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 1.14 1.15 1.16 1.17 Patibandla, S; Heaton, J; Azzam, JS (January 2020). "Aortic Insufficiency". PMID 32491360.
{{cite journal}}: Cite journal requires|journal=(help) - ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 Flint, N; Wunderlich, NC; Shmueli, H; Ben-Zekry, S; Siegel, RJ; Beigel, R (3 June 2019). "Aortic Regurgitation". Current cardiology reports. 21 (7): 65. doi:10.1007/s11886-019-1144-6. PMID 31161305.
- ↑ Sebastian, Anton (2018). A Dictionary of the History of Medicine. Routledge. p. PT540. ISBN 978-1-351-46999-9. Archived from the original on 2021-08-27. Retrieved 2020-12-21.
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 4.7 4.8 Chapter 1: Diseases of the Cardiovascular system > Section: Valvular Heart Disease in: Elizabeth D Agabegi; Agabegi, Steven S. (2008). Step-Up to Medicine (Step-Up Series). Hagerstwon, MD: Lippincott Williams & Wilkins. ISBN 978-0-7817-7153-5.
- ↑ Goudar, Raghavendra B.; ElBebawy, Bishoy (2023). "Pulsus Bisferiens". StatPearls. StatPearls Publishing. Archived from the original on 2023-06-30. Retrieved 2023-05-21.
- ↑ 6.0 6.1 6.2 6.3 6.4 6.5 "Aortic Regurgitation: Background, Pathophysiology, Etiology". 2018-11-19. Archived from the original on 2016-06-09. Retrieved 2016-06-02.
{{cite journal}}: Cite journal requires|journal=(help) - ↑ Schade R, Andersohn F, Suissa S, Haverkamp W, Garbe E (2007). "Dopamine agonists and the risk of cardiac-valve regurgitation". N. Engl. J. Med. 356 (1): 29–38. doi:10.1056/NEJMoa062222. PMID 17202453.
- ↑ Zanettini R, Antonini A, Gatto G, Gentile R, Tesei S, Pezzoli G (2007). "Valvular heart disease and the use of dopamine agonists for Parkinson's disease". N. Engl. J. Med. 356 (1): 39–46. doi:10.1056/NEJMoa054830. PMID 17202454.
- ↑ Hijazi, Ziyad M.; Ruiz, Carlos E.; Bonhoeffer, Philipp; Feldman, Ted (2006-01-17). Transcatheter Valve Repair. CRC Press. p. 31. ISBN 9781841844725. Archived from the original on 2021-08-27. Retrieved 2019-01-07.
- ↑ 10.0 10.1 10.2 Maurer, Gerald (2006-07-01). "Aortic regurgitation". Heart. 92 (7): 994–1000. doi:10.1136/hrt.2004.042614. ISSN 1355-6037. PMC 1860728. PMID 16775114.
- ↑ Galbraith, Alan; Bullock, Shane; Manias, Elizabeth; Hunt, Barry; Richards, Ann (2015-08-12). Fundamentals of Pharmacology: An Applied Approach for Nursing and Health. Routledge. p. 483. ISBN 9781317325871.
- ↑ Ginsburg, Geoffrey S.; Willard, Huntington F. (2013-01-01). Genomic and Personalized Medicine. Academic Press. p. 543. ISBN 9780123822277.
- ↑ Kumar, Vinay; Abbas, Abul K.; Aster, Jon C. (2012-05-01). Robbins Basic Pathology. Elsevier Health Sciences. p. 331. ISBN 978-1455737871.
- ↑ Topol, Eric J.; Califf, Robert M. (2007). Textbook of Cardiovascular Medicine. Lippincott Williams & Wilkins. p. 381. ISBN 9780781770125. Archived from the original on 27 August 2021. Retrieved 4 June 2016.
- ↑ Haase, Jürgen; Schäfers, Hans-Joachim; Sievert, Horst; Waksman, Ron (2010-04-01). Cardiovascular Interventions in Clinical Practice. John Wiley & Sons. p. 104. ISBN 9781444316711.
- ↑ Haase, Jürgen; Schäfers, Hans-Joachim; Sievert, Horst; Waksman, Ron (2010-04-01). Cardiovascular Interventions in Clinical Practice. John Wiley & Sons. p. 102. ISBN 9781444316711. Archived from the original on 2021-08-27. Retrieved 4 June 2016.
- ↑ "Aortic Valve Anatomy: Overview, Gross Anatomy, Microscopic Anatomy". 2018-09-25. Archived from the original on 2016-06-01. Retrieved 2016-06-04.
{{cite journal}}: Cite journal requires|journal=(help) - ↑ Mulroney, Susan; Myers, Adam (2015-08-31). Netter's Essential Physiology. Elsevier Health Sciences. p. 118. ISBN 9780323375849.
- ↑ Mandeville, Lisa K.; Troiano, Nan H. (1999-01-01). High Risk and Critical Care Intrapartum Nursing. Lippincott Williams & Wilkins. p. 67. ISBN 9780397554676.
- ↑ "UOTW#69 - Ultrasound of the Week". Ultrasound of the Week. 3 April 2016. Archived from the original on 15 December 2016. Retrieved 27 May 2017.
- ↑ Lancellotti, P.; Tribouilloy, C.; Hagendorff, A.; Moura, L.; Popescu, B. A.; Agricola, E.; Monin, J. L.; Pierard, L. A.; Badano, L.; Zamorano, J. L.; Sicari, R.; Vahanian, A.; Roelandt, J. R. T. C. (7 April 2010). "European Association of Echocardiography recommendations for the assessment of valvular regurgitation. Part 1: aortic and pulmonary regurgitation (native valve disease)" (PDF). European Journal of Echocardiography. 11 (3): 223–244. doi:10.1093/ejechocard/jeq030. PMID 20375260. Archived (PDF) from the original on 5 August 2016. Retrieved 4 June 2016.
- ↑ 22.0 22.1 Bonow, RO; American Heart Association Task Force on Practice Guidelines (Writing Committee to revise the 1998 guidelines for the management of patients with valvular heart disease); Society of Cardiovascular Anesthesiologists; Bonow, RO; Carabello, BA; Chatterjee, K; De Leon Jr, AC; Faxon, DP; Freed, MD (2006). "ACC/AHA guidelines for the management of patients with valvular heart disease. A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines". J. Am. Coll. Cardiol. 48 (3): e1–148. doi:10.1016/j.jacc.2006.05.021. PMID 16875962.
{{cite journal}}: Unknown parameter|displayauthors=ignored (help) - ↑ Nishimura, Rick A.; Otto, Catherine M.; Bonow, Robert O.; Carabello, Blase A.; Erwin, John P.; Guyton, Robert A.; O'Gara, Patrick T.; Ruiz, Carlos E.; Skubas, Nikolaos J. (2014-07-01). "2014 AHA/ACC guideline for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines". The Journal of Thoracic and Cardiovascular Surgery. 148 (1): e1–e132. doi:10.1016/j.jtcvs.2014.05.014. ISSN 1097-685X. PMID 24939033.
- ↑ Bickley, Lynn S.; Szilagyi, Peter G.; Bates, Barbara (2009-01-01). Bates' Guide to Physical Examination and History Taking. Lippincott Williams & Wilkins. p. 368. ISBN 9780781780582.
- ↑ Babu AN, Kymes SM, Carpenter Fryer SM (2003). "Eponyms and the diagnosis of aortic regurgitation: what says the evidence?". Ann. Intern. Med. 138 (9): 736–42. doi:10.7326/0003-4819-138-9-200305060-00010. PMID 12729428.
- ↑ Tang, Hong; Zhang, Jinhui; Sun, Jian; Qiu, Tianshuang; Park, Yongwan (1 April 2016). "Phonocardiogram signal compression using sound repetition and vector quantization". Computers in Biology and Medicine. 71: 24–34. doi:10.1016/j.compbiomed.2016.01.017. ISSN 0010-4825. PMID 26871603.
- ↑ Murthy, Pothuri Radha Krishna (2013-07-30). Heart in Fours: Cardiology for Residents and Practitioners. JP Medical Ltd. p. 47. ISBN 9789350904930. Archived from the original on 2021-11-10. Retrieved 4 June 2016.
- ↑ 28.0 28.1 28.2 28.3 Camm, Christian F.; Camm, A. John (2016-01-06). Clinical Guide to Cardiology. John Wiley & Sons. p. 10. ISBN 9781119079255. Archived from the original on 2021-11-10. Retrieved 4 June 2016.
- ↑ 29.0 29.1 29.2 29.3 29.4 29.5 29.6 29.7 29.8 Ashrafian, Hutan (8 March 2006). "Pulsatile pseudo-proptosis, aortic regurgitation and 31 eponyms". International Journal of Cardiology. 107 (3): 421–423. doi:10.1016/j.ijcard.2005.01.060. ISSN 0167-5273. PMID 16503268. – via ScienceDirect (Subscription may be required or content may be available in libraries.)
- ↑ Mokadam, Nahush A.; Stout, Karen K.; Verrier, Edward D. (2011-01-01). "Management of Acute Regurgitation in Left-Sided Cardiac Valves". Texas Heart Institute Journal. 38 (1): 9–19. ISSN 0730-2347. PMC 3060740. PMID 21423463.
- ↑ Stout, Karen K.; Verrier, Edward D. (2009-06-30). "Acute Valvular Regurgitation". Circulation. 119 (25): 3232–3241. doi:10.1161/CIRCULATIONAHA.108.782292. ISSN 0009-7322. PMID 19564568.
- ↑ Bekeredjian, Raffi; Grayburn, Paul A. (2005-07-05). "Valvular Heart Disease Aortic Regurgitation". Circulation. 112 (1): 125–134. doi:10.1161/CIRCULATIONAHA.104.488825. ISSN 0009-7322. PMID 15998697.
- ↑ "Aortic Regurgitation. Health Information and treatment | Patient". Patient. Archived from the original on 2016-06-18. Retrieved 2016-06-04.
- ↑ "Heart Failure Medication: Beta-Blockers, Alpha Activity, Beta-Blockers, Beta-1 Selective, ACE Inhibitors, ARBs, Inotropic Agents, Vasodilators, Nitrates, B-type Natriuretic Peptides, I(f) Inhibitors, ARNIs, Diuretics, Loop, Diuretics, Thiazide, Diuretics, Other, Diuretics, Potassium-Sparing, Aldosterone Antagonists, Selective, Alpha/Beta Adrenergic Agonists, Calcium Channel Blockers, Anticoagulants, Cardiovascular, Opioid Analgesics". emedicine.medscape.com. Archived from the original on 2016-06-05. Retrieved 2016-06-04.
- ↑ 35.0 35.1 Wilson W, Taubert KA, Gewitz M, et al. (October 2007). "Prevention of Infective Endocarditis: Guidelines from the American Heart Association". Circulation. 116 (15): 1736–54. doi:10.1161/CIRCULATIONAHA.106.183095. PMID 17446442.
- ↑ Picano, Eugenio; Pibarot, Philippe; Lancellotti, Patrizio; Monin, Jean Luc; Bonow, Robert O. (2009-12-08). "The Emerging Role of Exercise Testing and Stress Echocardiography in Valvular Heart Disease". Journal of the American College of Cardiology. 54 (24): 2251–2260. doi:10.1016/j.jacc.2009.07.046. ISSN 0735-1097. PMID 19958961. Archived from the original on 2016-06-11. Retrieved 2016-06-04.
- ↑ Members, Committee; Klocke, Francis J.; Baird, Michael G.; Lorell, Beverly H.; Bateman, Timothy M.; Messer, Joseph V.; Berman, Daniel S.; O’Gara, Patrick T.; Carabello, Blase A. (2003-09-16). "ACC/AHA/ASNC Guidelines for the Clinical Use of Cardiac Radionuclide Imaging—Executive Summary A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (ACC/AHA/ASNC Committee to Revise the 1995 Guidelines for the Clinical Use of Cardiac Radionuclide Imaging)". Circulation. 108 (11): 1404–1418. doi:10.1161/01.CIR.0000080946.42225.4D. ISSN 0009-7322. PMID 12975245.
- ↑ Choices, NHS. "Aortic valve replacement - Why it's done - NHS Choices". www.nhs.uk. Archived from the original on 7 June 2016. Retrieved 4 June 2016.
- ↑ 39.0 39.1 Bonow, Robert O. (2013-02-19). "Chronic Mitral Regurgitation and Aortic Regurgitation". Journal of the American College of Cardiology. 61 (7): 693–701. doi:10.1016/j.jacc.2012.08.1025. ISSN 0735-1097. PMID 23265342. Archived from the original on 2016-06-11. Retrieved 2016-06-04.
- ↑ Carrel, Thierry (2009-01-01). "Aortic valve and/or aortic root replacement using an aortic homograft". Multimedia Manual of Cardio-Thoracic Surgery. 2009 (626): mmcts.2009.003905. doi:10.1510/mmcts.2009.003905. ISSN 1813-9175. PMID 24413404.
- ↑ Prendergast, Bernard D.; Tornos, Pilar (2010-03-09). "Surgery for Infective Endocarditis Who and When?". Circulation. 121 (9): 1141–1152. doi:10.1161/CIRCULATIONAHA.108.773598. ISSN 0009-7322. PMID 20212293.
- ↑ "Aortic Regurgitation / Aortic insufficiency information. Patient | Patient". Patient. Archived from the original on 2016-05-24. Retrieved 2016-06-02.
Further reading
- Hamirani, Yasmin S.; Dietl, Charles A.; Voyles, Wyatt; Peralta, Mel; Begay, Darlene; Raizada, Veena (2012-08-28). "Acute Aortic Regurgitation". Circulation. 126 (9): 1121–1126. doi:10.1161/CIRCULATIONAHA.112.113993. ISSN 0009-7322. PMID 22927474.
External links
| Classification | |
|---|---|
| External resources |
- Pages with script errors
- CS1 errors: missing periodical
- CS1 errors: unsupported parameter
- Pages containing links to subscription-or-libraries content
- All articles with unsourced statements
- Articles with unsourced statements from June 2016
- Articles with invalid date parameter in template
- Valvular heart disease
- RTT
