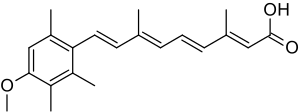
Acitretin
 | |
| Names | |
|---|---|
| Trade names | Soriatane, Neotigason, others |
| |
| Clinical data | |
| Drug class | Retinoid[1] |
| Main uses | Psoriasis, discoid lupus, ichthyosis, Darier's disease[2][1] |
| Side effects | Inflammation of the lips, hair loss, itchiness, dry eyes, joint pain, thinning of the skin[2] |
| Pregnancy category |
|
| Routes of use | By mouth |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601010 |
| Legal | |
| Legal status | |
| Pharmacokinetics | |
| Bioavailability | 60% |
| Protein binding | >99.9% |
| Metabolism | Hepatic |
| Elimination half-life | 49 hours |
| Excretion | Faeces & urine |
| Chemical and physical data | |
| Formula | C21H26O3 |
| Molar mass | 326.436 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Acitretin, sold under the trade name Soriatane among others, is a medication used to treat psoriasis, discoid lupus, ichthyosis, and Darier's disease.[2][1] Use is only recommended in severe disease were other treatments are not effective.[2] It is taken take by mouth.[2] Full benefits may take 2 to 3 months.[2]
Common side effects include inflammation of the lips, hair loss, itchiness, dry eyes, joint pain, and thinning of the skin.[2] Other side effects may include suicidal thoughts, liver problems, easy sunburns, and benign intracranial hypertension.[1] Use during, or the two years prior to pregnancy may harm the baby.[2] It is in the retinoid family of medications.[1] It is believed to work by prevention retinol being converted to retinoic acid.[2]
Acitretin was approved for medical use in the United States in 1996.[2] It is available as a generic medication.[3] In the United States a month of treatment costs about 90 USD.[3] In the United Kingdom this amount costs the NHS about 25 pounds as of 2021.[1] Following us a person should not giving blood for at least three years due to the risk of birth defects in those who receive the blood.[1]
Medical uses
It is used to treat psoriasis and discoid lupus.[2] Use is only recommended in severe disease were other treatments are not effective.[2]
-
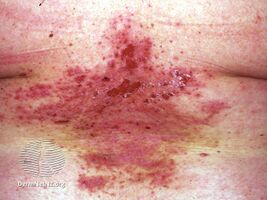
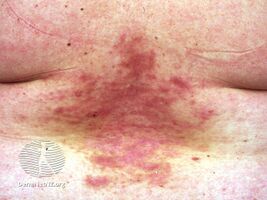
Darier disease before acitretin
-
Darier disease on acitretin
Dosage
The dose is 25 to 50 mg once per day.[2]
Side effects
Acitretin use during pregnancy is likely to cause severe birth defects. It should not be used by pregnant women or women planning to get pregnant within 3 years following the use of acitretin. Sexually active women of childbearing age who use acitretin should also use at least two forms of birth control concurrently. Men and women who use it should not donate blood for three years after using it, because of the possibility that the blood might be used in a pregnant patient and cause birth defects. In addition, it may cause nausea, headache, itching, dry, red or flaky skin, dry or red eyes, dry or chapped lips, swollen lips, dry mouth, thirst, cystic acne or hair loss.[4][5][6]
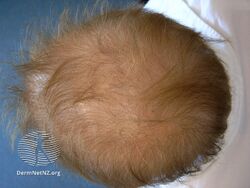
-
Acitretin diffuse hair loss
-
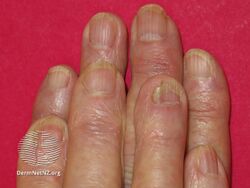
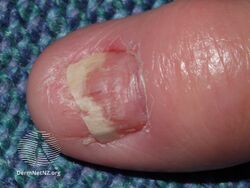
Acitretin nail thinning
-
Acitretin nail thinning
Pharmacokinetics
It is a metabolite of etretinate, which was used prior to the introduction of acitretin. Etretinate was discontinued because it had a narrow therapeutic index as well as a long elimination half-life (t1/2=120 days), making dosing difficult. In contrast, acitretin's half-life is approximately 2 days. However, because acitretin can be reverse metabolised into etretinate which has an extremely long half-life, women must avoid becoming pregnant for at least three years[7] after discontinuing acitrtin. Therefore, acitretin is generally not recommended for women of child-bearing age with a risk of becoming pregnant.[8]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 BNF (80 ed.). BMJ Group and the Pharmaceutical Press. September 2020 – March 2021. p. 1327. ISBN 978-0-85711-369-6.
{{cite book}}: CS1 maint: date format (link) - ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 2.11 2.12 "Acitretin Monograph for Professionals". Drugs.com. Archived from the original on 6 March 2021. Retrieved 18 July 2021.
- ↑ 3.0 3.1 "Acitretin Prices, Coupons & Savings Tips - GoodRx". GoodRx. Archived from the original on 19 September 2016. Retrieved 18 July 2021.
- ↑ "Soriatane". WebMD. Archived from the original on 2 December 2013. Retrieved 15 August 2015.
- ↑ "Soriatane Side Effects". Drugs.com. Archived from the original on 6 May 2018. Retrieved 15 August 2015.
- ↑ "Soriatane (Acitretin) Drug Information: Description, User Reviews, Drug Side Effects, Interactions - Prescribing Information at RxList". RxList. Archived from the original on 2 December 2013. Retrieved 15 August 2015.
- ↑ "Important Safety Information for SORIATANE". soriatane.com. Archived from the original on 6 May 2018. Retrieved 31 October 2015.
- ↑ Zito, Patrick M.; Mazzoni, Thomas (2022). "Acitretin". StatPearls. StatPearls Publishing. Archived from the original on 2022-12-23. Retrieved 2023-08-08.
External links
| Identifiers: |
|
|---|
- Pages using duplicate arguments in template calls
- CS1 maint: date format
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Retinoids
- Phenol ethers
- Enones
- Hoffmann-La Roche brands
- RTT